Introduction
Identifying the diversity of sensory neurons and their role in the sense of touch and pain is important for diagnosing and treating peripheral neuropathies, common disorders of the peripheral nervous system [1,2]. Understanding the connection between sensory deficit, chronic pain, and pathologic changes in sensory neurons could aid in the development of new pain therapies with the purpose of improving nervous system dysfunction. The results reported here show how rapid, high-contrast 3D imaging of sensory neurons with a THUNDER Imager can help researchers gain insights into neuropathies.
Challenges
A solution that can quickly achieve high-contrast imaging of thick ganglion tissue, where important specimen details are clearly resolved, is important for neuropathy research. Conventional widefield microscopy offers speed and detection sensitivity, however there is often an out-of-focus blur or haze in the images, due to signals from out-of-focus planes of thick tissues. These significantly reduce image contrast [3,4].
Methods
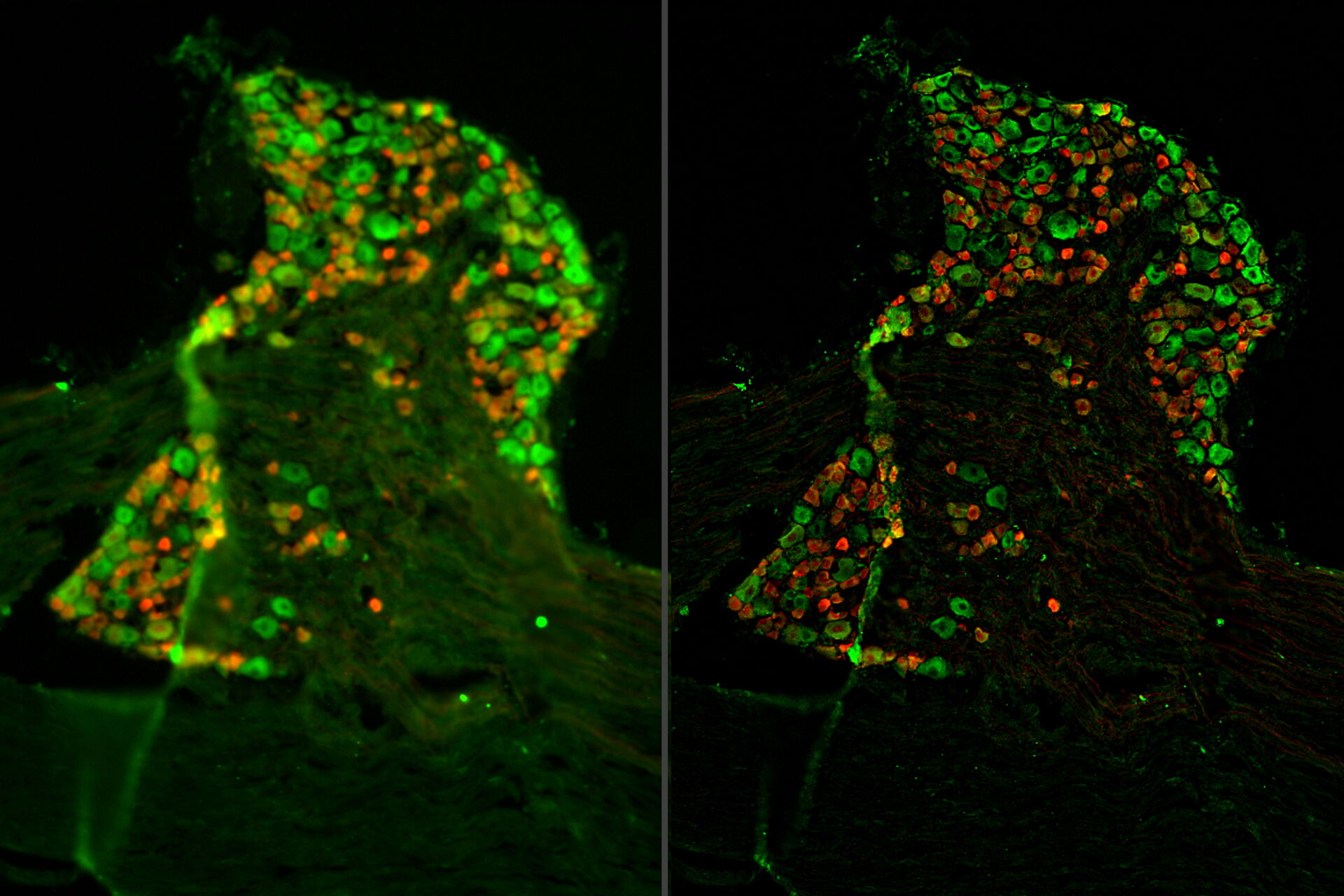
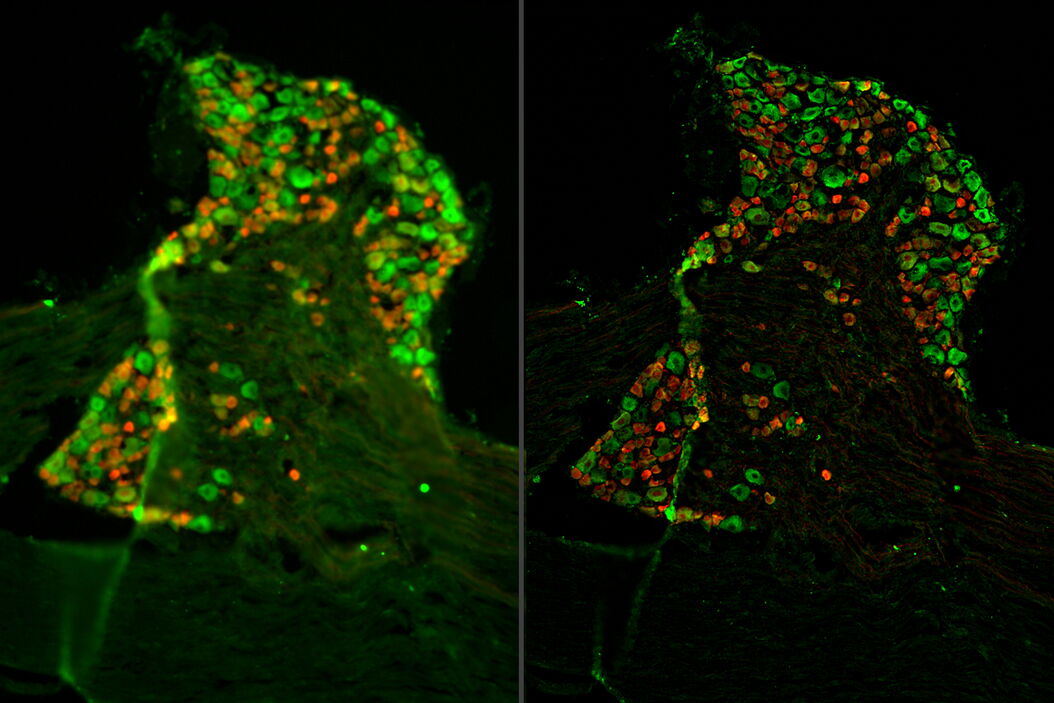
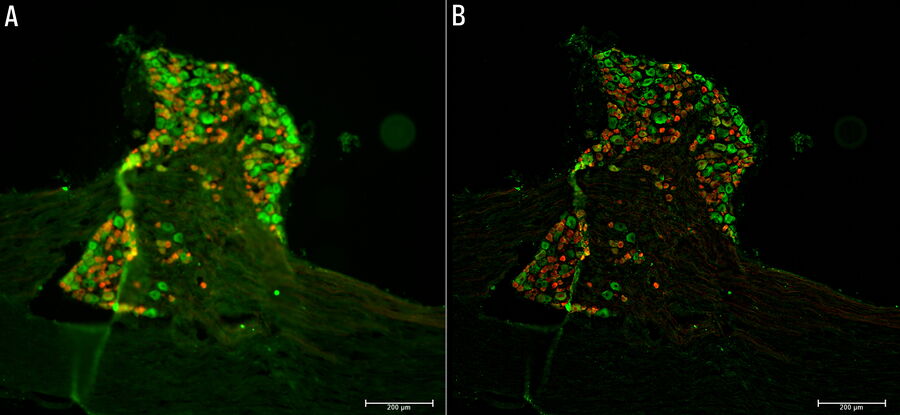
Tissue specimens of dorsal root ganglion (DRG) were prepared from a transgenic mouse expressing tdTomato (red) in a subset of sensory neurons. Paraformaldehyde-fixed, 20-μm thick cryosections were stained overnight with anti-mCherry primary antibody, then stained with Cy3-conjugated secondary antibody, and mounted with Fluoromount G. Background neuronal autofluorescence was detected by the GFP filter set to allow for neuronal counting (green). A three-dimensional z-stack of images was acquired with a THUNDER Imager Tissue and large volume computational clearing (LVCC) [3,4] was applied. A two-dimensional projection was created using the LAS X Extended Depth of Field processing tool.
Results
Images of the ganglion tissue specimen acquired with the THUNDER Imager are shown below in figure 1.
Conclusions
The THUNDER Imager Tissue and large volume computational clearing (LVCC) can bring clarity to widefield fluorescent images of the mouse tissue through the removal of out-of-focus blur, revealing fine structural details of the dorsal root ganglia.
References
- L.K. Crawford, M.J. Caterina, Functional Anatomy of the Sensory Nervous System: Updates From the Neuroscience Bench, Toxicologic Pathology (2020) vol. 48, iss. 1, pp. 174-189, DOI: 10.1177/0192623319869011.
- E.L. Tran, L.K. Crawford, Revisiting PNS Plasticity: How Uninjured Sensory Afferents Promote Neuropathic Pain, Front. Cell. Neurosci. (2020) vol. 14, DOI: 10.3389/fncel.2020.612982.
- J. Schumacher, L. Bertrand, THUNDER Technology Note: THUNDER Imagers: How Do They Really Work? Science Lab (2019) Leica Microsystems.
- L. Felts, V. Kohli, J.M. Marr, J. Schumacher, O. Schlicker, An Introduction to Computational Clearing: A New Method to Remove Out-of-Focus Blur, Science Lab (2020) Leica Microsystems.
Related Articles
-
What are the Challenges in Neuroscience Microscopy?
eBook outlining the visualization of the nervous system using different types of microscopy…
Jun 14, 2023Read article -
Central Nervous System (CNS) Development and Activity in Organisms
This article shows how studying central nervous system (CNS) development in Drosophila-melanogaster…
May 12, 2023Read article -
Going Beyond Deconvolution
Widefield fluorescence microscopy is often used to visualize structures in life science specimens…
Mar 22, 2023Read article
Related Pages
-

THUNDER Imaging Systems
To answer important scientific questions, THUNDER Imaging Systems enable you to obtain a clear view…
Visit related page