Physiology Images taken with THUNDER Imagers
See how Leica Microsystems instruments can help imaging cardiac organoids used to develop pacemaker cells for heart arrhythmias, muscular dystrophy, diabetes, sensory hair cell regeneration, cellular eye morphogenesis, retina and the vascular network, the Drosophila eye, HeLa cells, zebrafish heart, etc. There are different model organisms and specimens involved in this collection, such as chicken, mice, C. elegans, Drosophila, zebrafish, Arabidopsis thaliana, and HeLa cells.
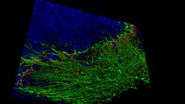
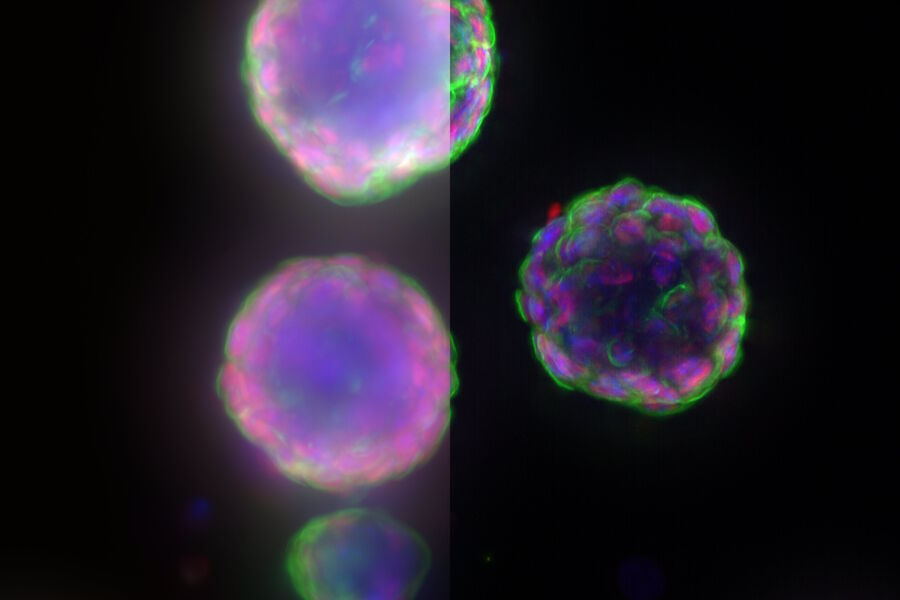
Cardiac Spheroids
Vesicle formation, movement, and fusion
This video shows how observation of intracellular membrane trafficking along microtubules is enhanced with THUNDER Computational Clearing. Shown is a time-lapse movie of living HeLa cells transfected with Rab5a-GFP and stained with SIR-Tubulin. Vesicle formation, movement, and fusion can be observed.
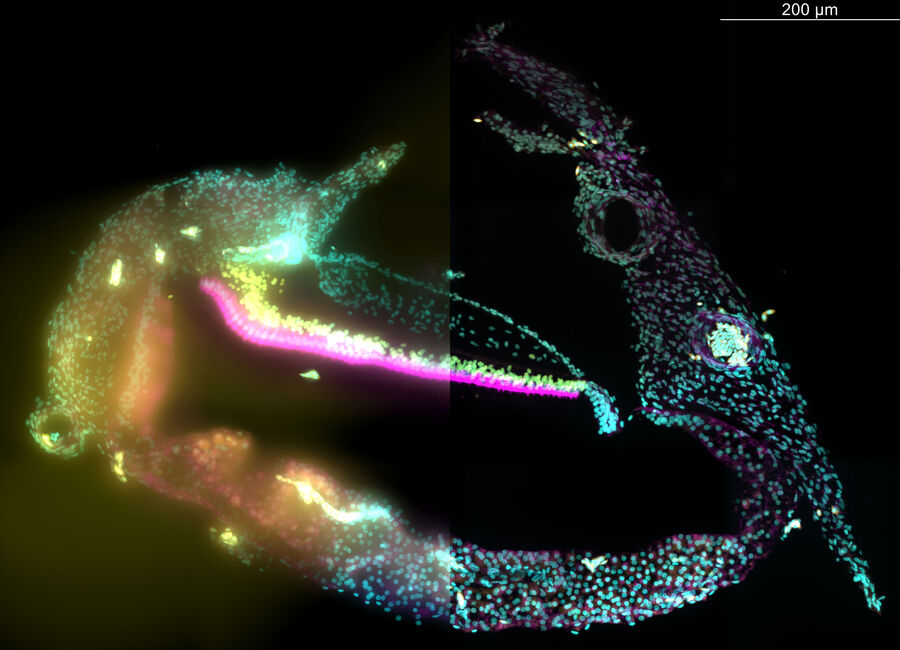
Zebrafish Heart
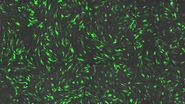
HeLa Cells
Mouse Retina
Dr. Jiyeon Lee, a scientist at Roche Genentech in South San Francisco, CA uses the mouse retina model to study the interaction between endothelial cells, blood vessels, and astrocytesin the retina. Dr. Lee uses whole mount retina preparation to visualize its vasculature. Following fixation and staining, the whole retina must be imaged at high resolution to provide an overview of the whole vascular network, as well as single cell interactions.

Arabidopsis Thaliana Root
Murine Esophageal Organoids
Adult Drosophila Muscle
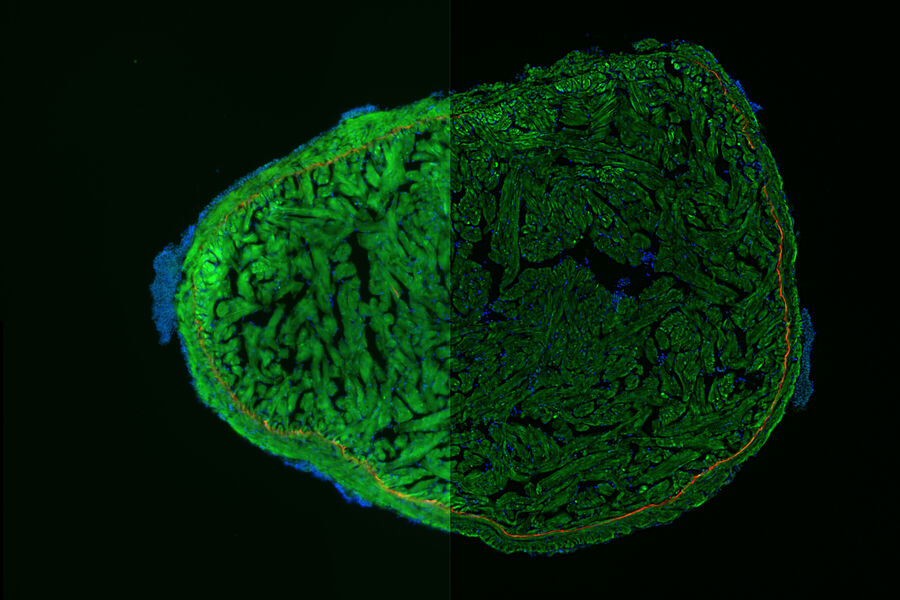
Mouse Lens Section
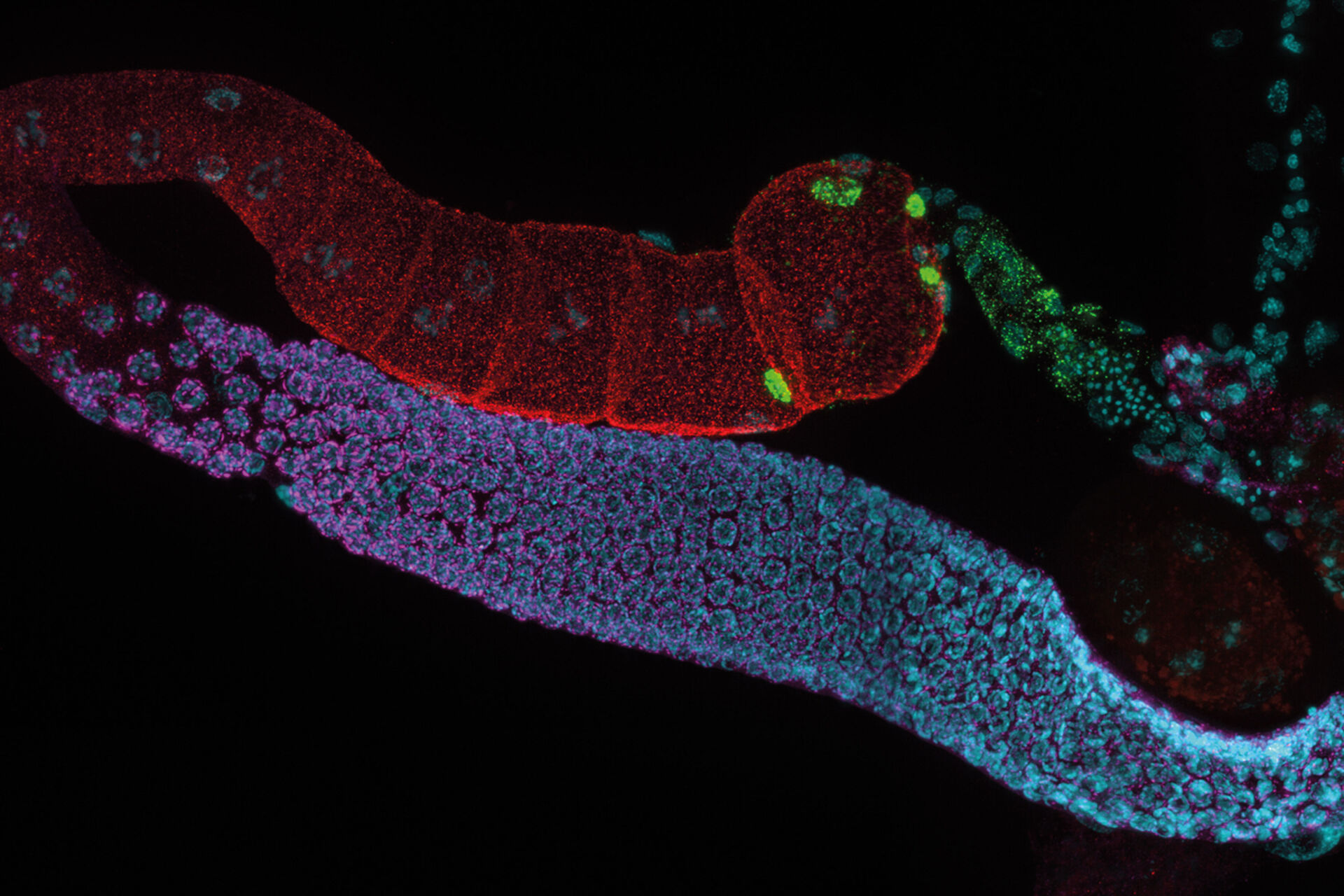
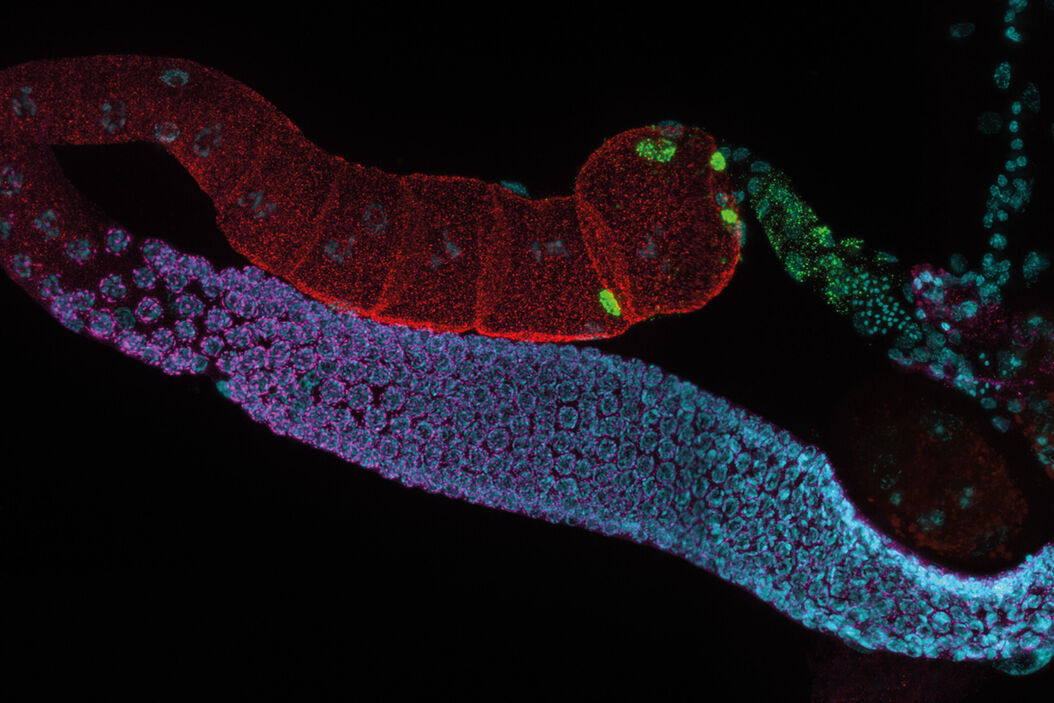
C. elegans Gonades
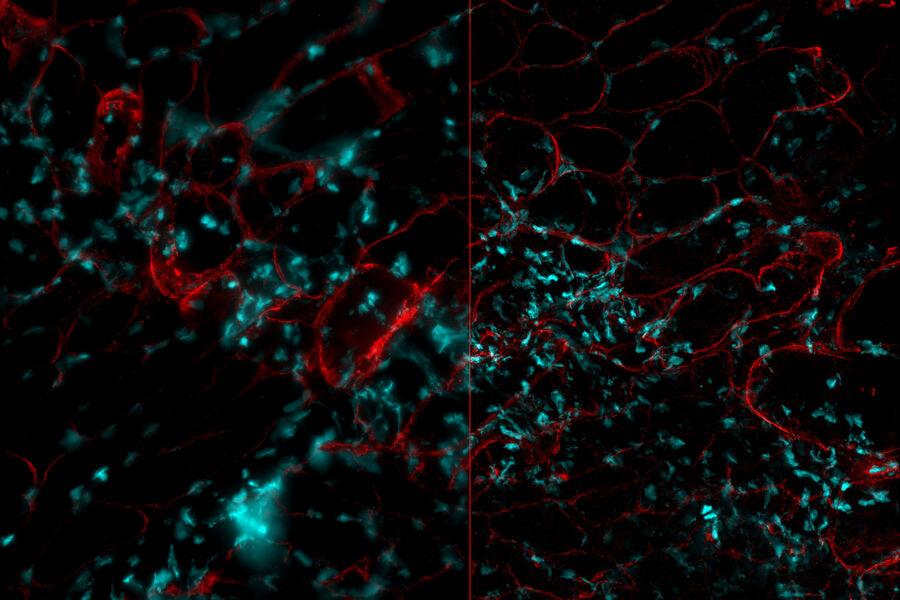
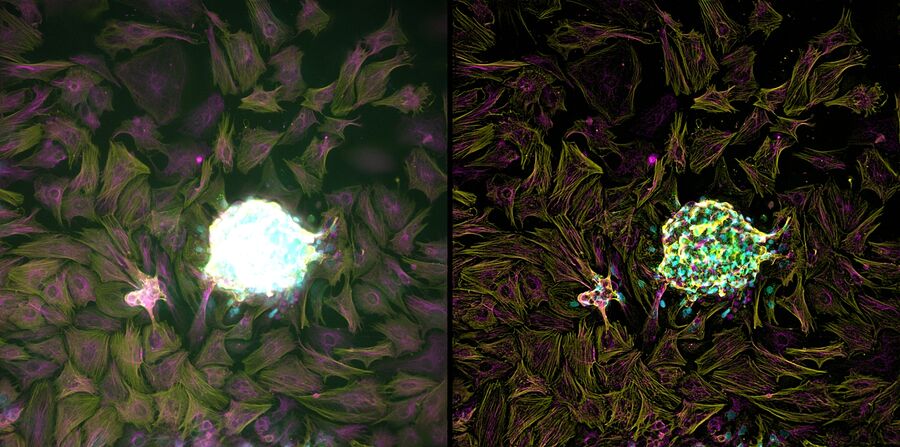
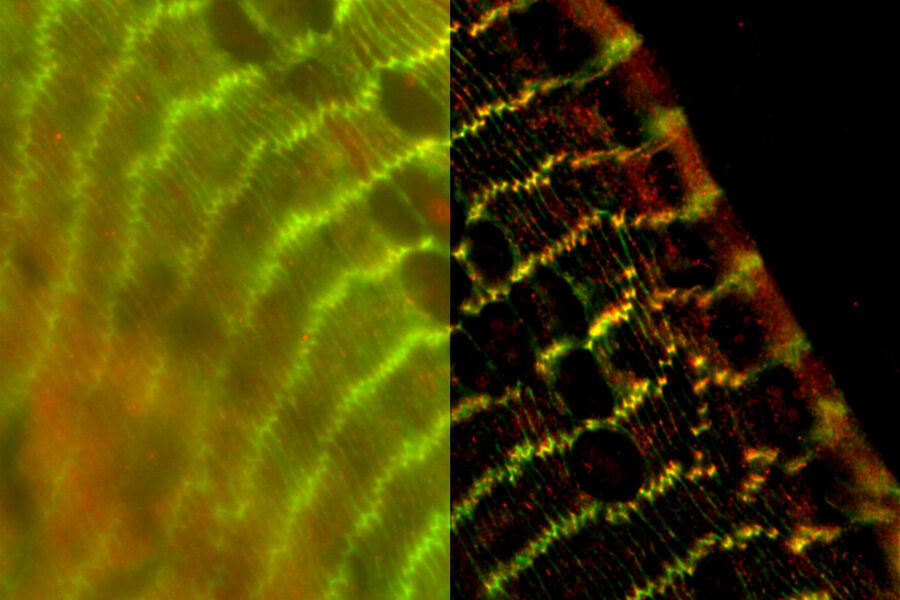
Chicken Cochlea Tilescan
Dr. Amanda Janesick, a postdoctoral fellow in the laboratory of Dr. Stefan Heller, studies the development and regeneration of the inner ear, specifically the sensory hair cells, which serve a mechanosensitive purpose in the inner ear. Dr. Janesick uses the chicken embryo as a model to study sensory hair cell regeneration, because of the ability of chickens to naturally recover from hearing loss.
Dr. Janesick uses thick vibratome sections of post-hatch 7 day chicken cochlear tissue to visualize the sensory hair cells in the inner ear. Typically, this is a challenging sample for widefield fluorescence microscopes, due to the haze inherent to thicker tissue samples, which obscures structures of interest (Figure 1, left panel). Using a THUNDER Imager 3D Tissue, the haze and background was cleared from this image using Instant Computational Clearing (ICC, Figure 1, right panel), making it suitable for observation of individual sensory hair cells (magenta) and supporting cells (yellow) of the inner ear. This 3-channel 10-position tilescan took under a minute to acquire and process with ICC. Because ICC is a 2D method, and does not require z-stacks, it could be applied to this single plane tilescan.
Transgenic Drosophila Photoreceptors of the Eye
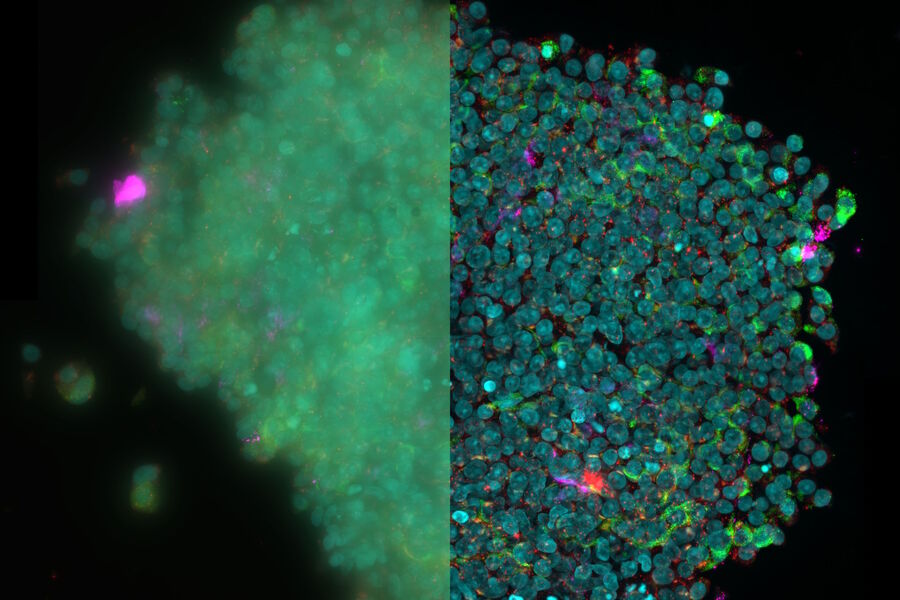
Pancreatic Islet
A better understanding of how type 1 diabetes (T1D) develops is the first step to potentially develop new therapies capable of preventing or permanently reversing the disease. Due to the inaccessibility to human pancreatic tissue, our knowledge of the disease in humans is limited. The Network for Pancreatic Organ donors with Diabetes (nPOD) was established with the idea of providing valuable tissues from healthy and diabetic donors toanswer basic questions about the pathogenesis of T1D. One of the interests of the von Herrath lab is toidentifycytokines in the pancreatic tissue samples of T1D cases obtained from nPOD. Since T1D is an autoimmune disease, where insulin producing pancreatic beta cells are attacked by the immune system, understanding the cytokine milieu in the pancreas of T1D will lead to a better understanding of the disease pathogenesis and contribute towards development of therapeutic targets for T1D.
Mouse Dystrophin Staining on Muscle Fibres