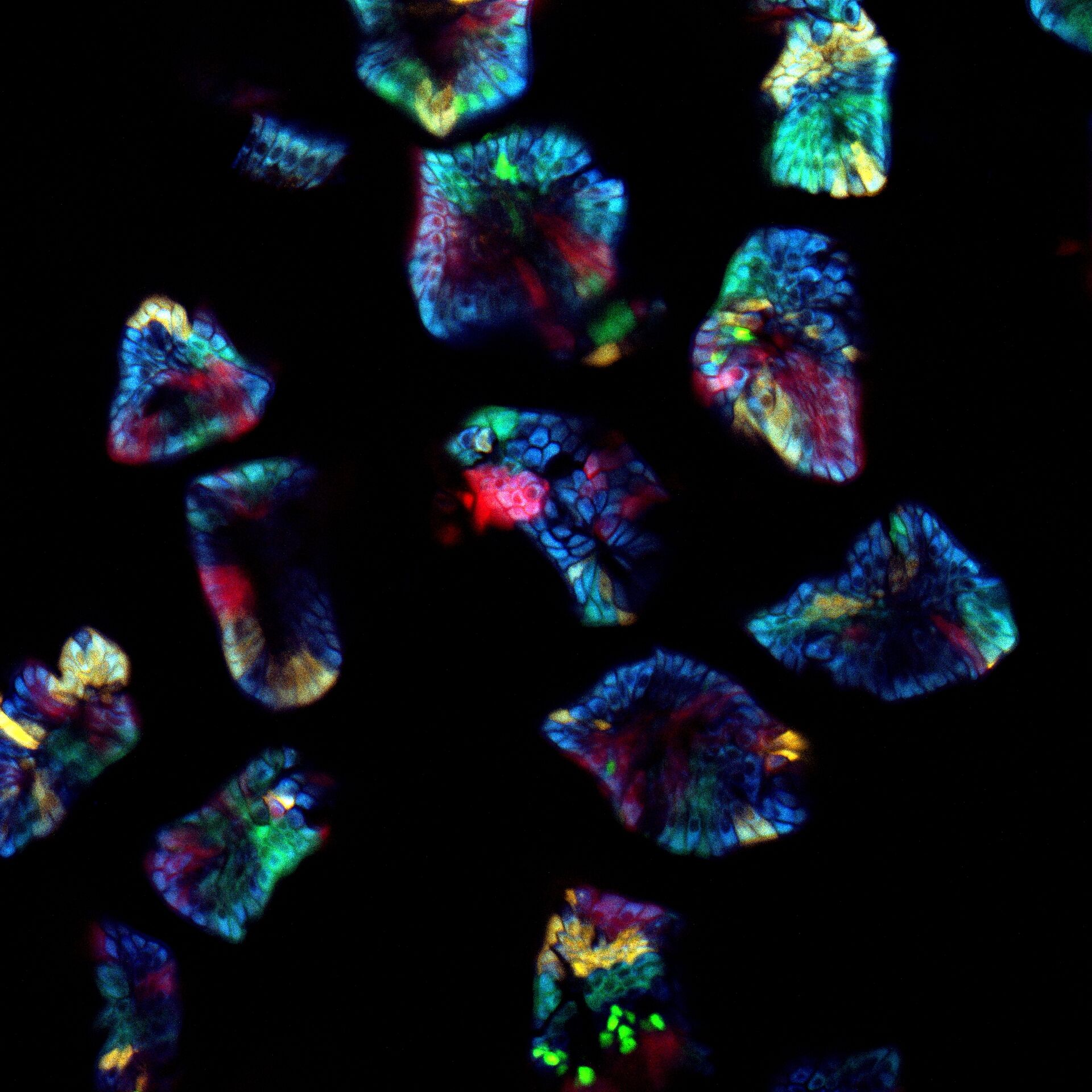
Nonlinear illumination
In 1931, while doing her doctoral thesis, Maria Göppert-Mayer postulated the excitability of an electronic system by the simultaneous absorption of two photons with half the energy needed for a transition from the ground state to an excited state [1]. If excitation of a dye is possible at, e.g. 450 nm, it should be possible to excite the same dye at around 900 nm as well. The critical word here is “simultaneously” which means a second photon must arrive at the excitable site when the first photon is still at that place. This phenomenon is achievable only if the spatial and temporal “concentration” of photons is very high, that is, at very high light intensities.
High densities of infrared light can be generated in the focal volume of an objective lens when illuminated with high intensity infrared (IR) lasers. To obtain a two-dimensional image, the focus must be scanned over the microscopic field. A scanning microscope for non-linear illumination was initially described by Colin Sheppard [2]. A full review of the topic of scanning confocal and multiphoton microscopy has been provided by A. Diaspro [3].
Why should one spend money for elaborate scanning microscopes and expensive IR-lasers, if fluorescence images can be obtained by simple fluorescence microscopes? Multiphoton excitation offers two significant benefits:
- Intrinsic optical sectioning:
The excitation depends on the square of the intensity for two-photon excited fluorescence. Sufficient intensity is only found in the focal volume. Consequently, only here molecules are excited and emit via fluorescence. - Illumination into deep layers:
Biological samples are turbid and cause Rayleigh-scattering that is inversely proportional to the fourth power of the wavelength. When the illumination wavelength is doubled, the losses by Rayleigh scattering shrink to 1/16th. (The subsequent emission of the fluorescent light and / or the higher order harmonics will still experience strong scattering.)
As the method is employing point-scanning microscopy, multiphoton excitation systems are often built on a platform of confocal beam scanning microscopes. The essential modification is the installation of high intensity pulsed IR-lasers.
Beam diameter
The main goal of microscopy is to provide magnified images with high optical resolution. The magnification is needed to adapt the resulting image to the resolution of our bodily viewing device, the human eye. The high optical resolution is required to reveal new fine details of the investigated object. Optical resolution is essentially ruled by the wavelength (color) of the light and by the numerical aperture of the objective lens in use. The theoretical limit is reached when the optical device can separate two objects spaced by a distance d defined as: dA = λ/2NA.
This formula resembles the considerations of Ernst Abbe [4][5], indicated by the subscript A. Depending on the methods and preconditions, the theoretical limit is different, but always not too far away from the Abbe-formula. (For a brief comparison refer to another report by the author [6]).
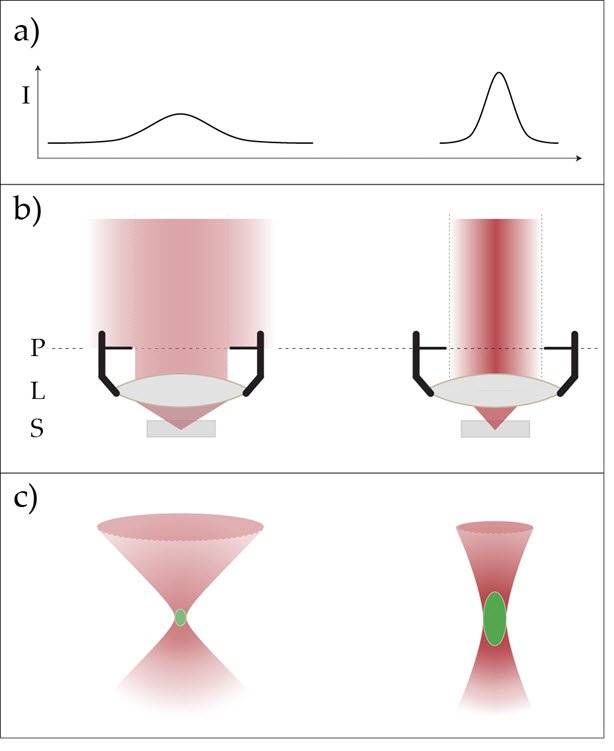
This limit requires the illumination to be homogeneously distributed in the back focal plane of the objective lens. That requirement means that the intensity is even: identical brightness at every point of the pupil plane (Figure 1).
The intensity across a laser beam (cross section beam width) is not even. It rather resembles a 2D-Gaussian distribution (in the best case) (Figure 1a). To ensure homogeneous illumination, the laser beam usually is widened and only the inner fraction is used. The residual decrease of intensity at the periphery is then negligible (Figure 1, left row).
However, this manipulation of the beam causes a technical dilemma: for highest resolution, the intensity must be even, but for best multiphoton excitation deep inside the sample we need the highest intensities possible and cannot afford to cut away significant parts of the laser beam’s cross section [7]. The practical solution is a variable beam expander (VBE) allowing the user to decide which parameter for the given experimental task is most important and adapt the beam diameter correspondingly (Figure 1). The optimal beam diameter depends on the size of the pupil and should be adapted accordingly.
The VBE from Leica Microsystems is a tunable optical beam expander, allowing the user to modify the illumination beam diameter over a wide range to fulfill the requirements for resolution and penetration depth. The module is capable of holding up to four individual variable beam expander optics that can control the performance of up to four different IR wavelengths individually and simultaneously.
Focus position
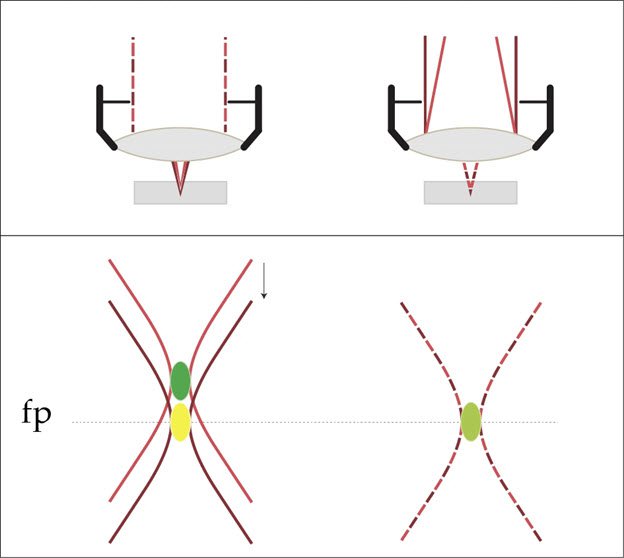
Due to the wavelength-dependence of refraction, rays of different color will show different focal positions when passing through a collecting lens. This effect is known as axial color aberration. For the visible range of the electromagnetic spectrum, from 400 to 700 nanometers, optical lenses for microscopes and cameras are designed to minimize this type of aberration by combination of lenses that show different refractive characteristics. The better the correction, the higher the effort and the price. For special applications, lenses corrected in the near ultraviolet, as well as lenses that cover the near infrared range, are available.
Common lenses are not corrected for the use of several infrared wavelengths at the same time. Here, we can make use of the single color of laser beams and correct the excitation focus by means of variable optics. By introduction of a divergence bias, the focus may be shifted continually and adaptable to any infrared color. This result is achieved by exploiting the design of the variable beam expander, which offers two degrees of freedom that can be adjusted to control both the beam diameter and the focus position independently.
With that approach, it is possible to ensure different IR-wavelengths to be focused at the same position. A crucial tool for multiple IR-excitation to keep all images aligned in the z-direction.
The VBE offers beam shaping for up to four different IR illumination wavelengths. All four IR beams can be adjusted for resolution and penetration depth and independently for focus correction assuring a perfect coincidence for excitation with different IR colors.
References
- Göppert-Mayer M (1931) Über Elementarakte mit zwei Quantensprüngen. Göttinger Dissertationen. Annalen der Physik 401/3: 273–294
- Sheppard CJR and Kompfner R (1978) Resonant scanning optical microscope. Applied Optics 17/18 pp 2879-2882
- Diaspro A (Ed) (2002) Confocal and Two-Photon Microscopy. Wiley-Liss, New York
- Abbe EK (1873) Beiträge zur Theorie des Mikroskops und der mikroskopischen Wahrnehmung. Archiv für Mikroskopische Anatomie 9(1):413–468.
- Wilson M: Microscope Resolution: Concepts, Factors and Calculation (2016)
- Borlinghaus RT (2017) The White Confocal – Microscopic Optical Sectioning in all Colors. Springer International Publishing. Cham, Switzerland.
- Helmchen F and Denk W: Deep tissue two-photon microscopy. Nature Methods 2/12 (2005) 932-940