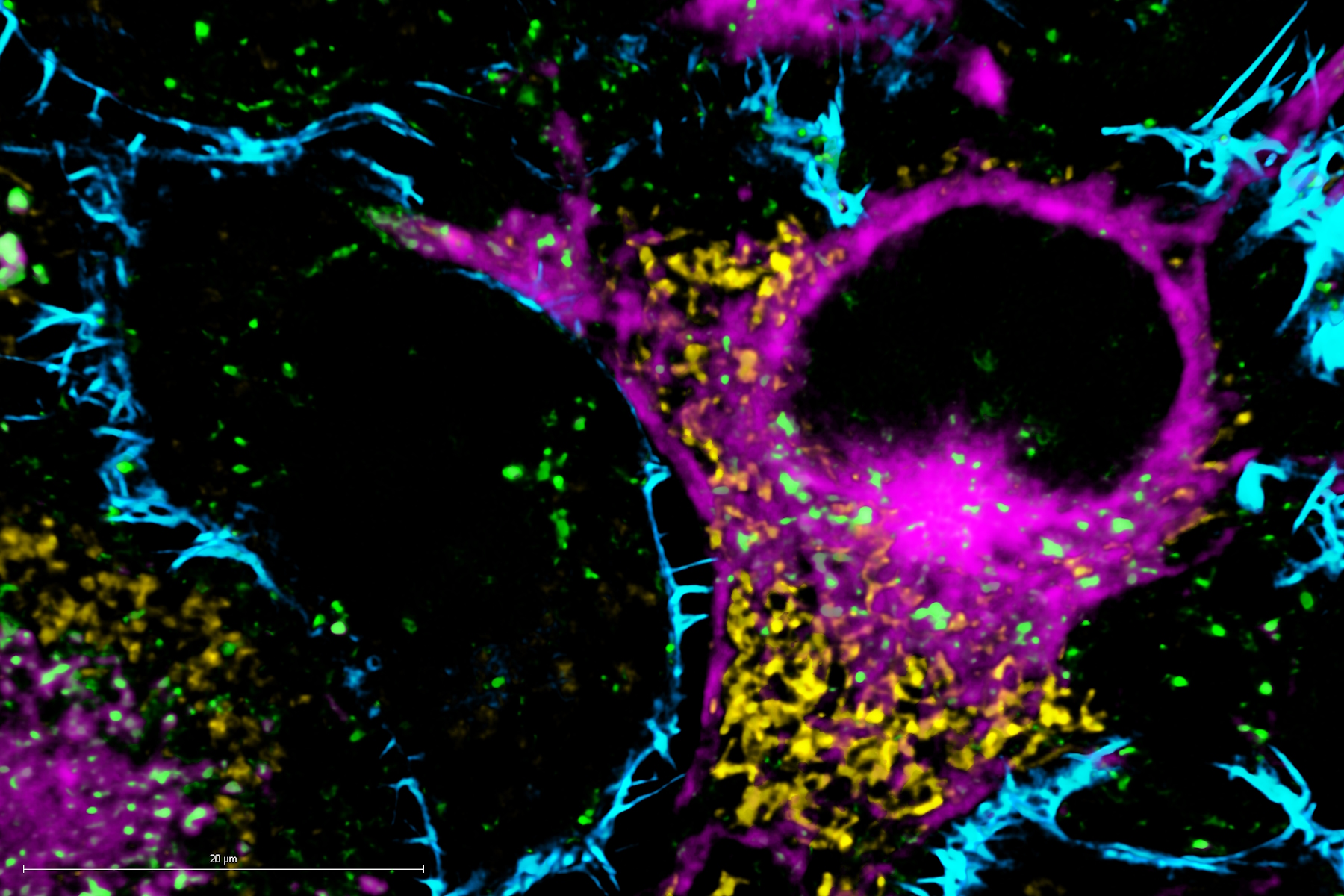
The supercontinuum white light source
Classical laser sources usually only provide one single narrow emission. Some gas lasers can emit a couple of lines simultaneously. The best known example is the argon gas laser, which may be available with 5 lines in the blue-green range. Other lasers, like dye lasers, are tunable, but emit only a single color at a time. In addition, dye lasers are very unstable and require some patience to run properly. There are solid state lasers that are tunable, but only for the IR range (e.g. Ti:Sa lasers). Beside their high complexity and expensiveness, they too can emit only a single wavelength at a time and the tuning process is very slow (many seconds).
This situation has entirely changed with the invention of light sources that emit over a broad range simultaneously, but still behave like lasers in terms of focusability [1].
These sources employ a fiber IR laser which emits light pulses at some 80 MHz. This compound is used as a "seed", i.e. provides a precise clock but has quite low energy. The pulses are subsequently amplified in a diode-pumped laser amplifier. The amplification laser is also fiber based, and both systems are coupled seamlessly by fiber splicing. The output of the amplifier may reach some 10 W of IR light average power, but chopped in pulses of ca. 200 ps at 80 MHz. These high energy pulses are finally focused on the entry surface of a so-called photonic crystal fiber (PCF). These fibers feature a pattern of hollow tubes in the center of the fiber. The strongly nonlinear processes at the surfaces in the tube pattern that cause monochromatic light to be spread into a broad spectrum reaching blue or even ultraviolet include group velocity dispersion (up to 4th order), self-steepening, Raman scattering and plasmon fission.
Depending on the length of the PCF, the monochromatic IR peak is spread to a spectrum of up to several hundreds of nanometers. Typical fiber lengths are 0.5 to 2 m.
Two-dimensional acousto-optical tuning of color and strength
Like in any other microscope that uses a white source for fluorescence illumination, the white light laser confocal microscope [2] requires a means to select colors to specifically excite various fluorochromes. If possible, a multitude of colors is useful for parallel excitation of multiply stained samples. In a widefield fluorescence microscope, this task is performed by the excitation filter. Dichroitic filters with three or four transmission bands are used for multi-stained samples.
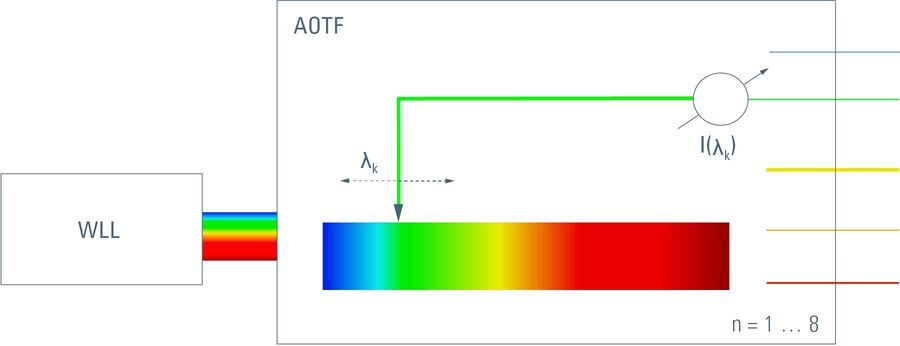
The beam emitted from a supercontinuum offers a more versatile solution. An acousto-optical tunable filter can pick a single line (which is actually a narrow bandlet) out of the spectrum and deflect it to a different direction. The rest of the spectrum will go straight through the crystal. As the color of the bandlet is steplessly tunable by the mechanical excitation frequency that controls the deflection, any color from the input spectrum is selectable. The width of that bandlet is some 1 … 3 nm, depending on the wavelength. The energy in such a bandlet is in the range of a few milliwatts, which is by far enough for imaging and frequently used even for FRAP experiments. By adding more electronic drivers to generate mechanical waves in the crystal, it is possible to direct a series of colors simultaneously into the 1st order. Eight colors simultaneously is standard (but not the technical limit). Each of these bandlets is tunable both in wavelength and in intensity.
This combination makes the white light laser (WLL) an ideal source for excitation in confocal microscopes. It will serve for all known dyes excitable in the visible range, it allows recording of excitation spectra to explore the spectral properties of new dyes, especially in situ. And it gives the freedom to tune the excitation off-peak if this is necessary for emission collection reasons or to improve separation of adjacent dyes.
Only acousto-optical beamsplitting (AOBS) can do the job
The very beneficial aspect of free tunability of the excitation color initiates a new challenge: what device can feed the steplessly tuned light into the beam path of an incident-light fluorescence microscope? The simplest approach would be to use a grey beam splitter, but this would be at the expense of always losing a certain fraction of the excitation energy – although this is the lesser problem. A severe drawback of such an approach is the loss of precious emission light. Even with an 80/20 grey splitter (which would always waste 4/5 of the laser energy) one fifth of the emission would be sacrificed.
A single dichroic mirror, even with multiple bands, could clearly not support the benefits of a tunable light source. An approach for a single-emission slow tuning, non-continuous light source has been commercialized. This system employs a sequence of ca. 10 dichroic mirrors that would be used at their operating frequency – giving only 10 colors out of a range of 200 – and they are also used off the specified wavelength at both sides. This makes a total of 30 colors, but with clearly less than perfect performance.
A perfect remedy for this predicament is to use acousto-optics. The acousto-optical tunable beam-splitter (AOBS) [3]is a device that allows a series of color bandlets to be fed into an optical path while the rest of the spectrum is guided into a different direction with over 95 % efficiency. In the case of a multi-bandlet tunable laser, one would operate the AOBS so that the excitation colors pass the 1st order as entry into the excitation beam path.
The complete emission spectrum will then be efficiently transmitted into the detection path. The interstices in the transmission spectrum required to thread the excitation bandlet onto the sample are the same size as the bandlet itself (as they are generated by identical technologies) and can be controlled in their position simultaneously: as soon as one tunes the excitation color, the corresponding interstice is tuned appropriately by the electronics in parallel. No action by the operator is required.
The Acousto-Optical tunable Beam Splitter is therefore the ultimate coupling device for tunable multicolor excitation.
Excitation and emission spectra of intensity and fluorescence lifetime
The spectral detector (SP) is designed for full control over the bandwidth and central color of the detected emission bands. This guarantees highest throughput (transmission efficiency) and optimal separation. As a side-effect, the SP detector allows recording of emission spectra as well – either simultaneously (5 channels) or sequentially (up to 400 channels), or in a mixed mode of both approaches. In the past, the emission color was bound to a few lines emitted by the lasers installed in a system. With the white light laser (WLL), the situation has changed entirely: as the color is tunable with 1nm accuracy, a smooth excitation spectrum is available throughout the visible spectrum. In combination with the SP detector, which can adapt servo-controlled to the excitation wavelength, this measurement is performed fully automatically. And it is only reasonably practicable by implementing the acousto-optical beam splitter to guide excitation and emission light.
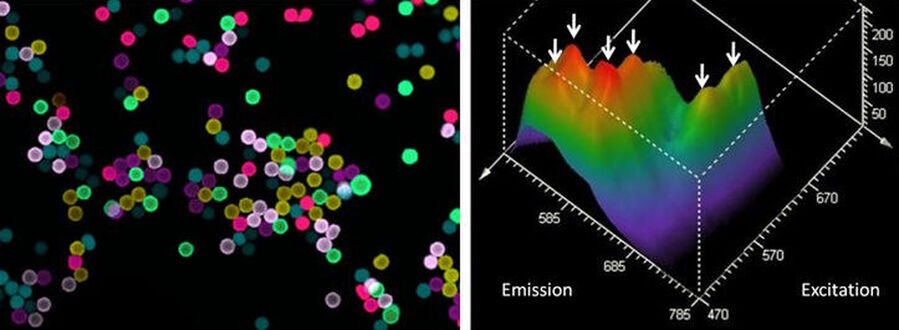
Having both a tunable excitation and a tunable emission, the next step is to create a two-dimensional correlation map of the intensities [4]. This lambda-square map is especially helpful when complex mixtures of fluorochromes need to be separated. It is also of general value when fluorochromes need to be characterized – either because they are newly found or designed, or because of an interest in spectral alterations due to microenvironmental conditions.
Fluorescence is characterized by emission intensity and by the fluorescence lifetime. To measure lifetime exactly, a pulsed light source is required. Fortunately, the white light laser is a pulsed source, and the pulse frequency fits the needs for typical fluorescence lifetimes (which are in the range of 0.5 … 5 nanoseconds). If longer pulse intervals are required, a pulse picker reduces the pulse frequency. And of course, the tunability of the white light laser directly renders lifetime excitation spectra possible, including the next logical consequence: a two-dimensional excitation-emission lifetime map.
The light gate – a panchromatic electronic barrier filter
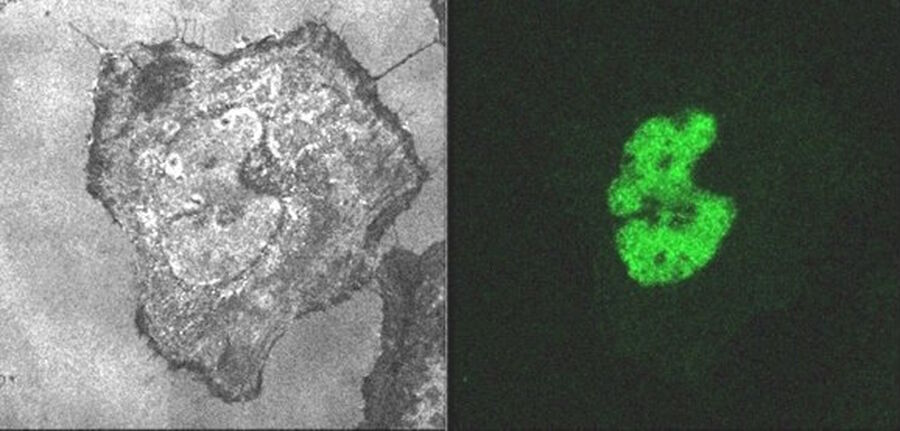
There is one last thing the latest technologies have enabled. Hybrid detectors (HyDs) are very sensitive, fast and cover a large dynamic range. They provide a gated operation mode as well. In combination with the pulsed white light laser, it is now possible to detect fluorescence emission only during the time between the light pulses – just by gating the hybrid detector to collect no signal during the laser pulse. The detected signal is then pure fluorescence emission and does not contain any reflected light. With that approach, the color battle against residual reflection is easily won. There is no need to restrict the emission collection by leaving sufficient space to the excitation color. It is even possible to measure directly underneath the excitation – and beyond, which then allows characterization of anti-Stokes emission.
The pulsed and spectrally tunable white light laser source, in combination with the tunable acousto-optical beam splitter, the tunable SP detector and the gated hybrid detectors is a fascinating blend of latest technologies. And, although the phrase may sound a little well-worn, it fits here: it revolutionizes the conception and operation of confocal microscopy.
References
- Knight JC, Birks TA, Russell TS and Atkin DM: All-silica single-mode optical fiber with photonic crystal cladding. Opt. Lett 21 (1996) 1547–49.
- Birk H and Storz R: Illuminating device and microscope US Pat. 6,611,643 (2001).
- Birk H, Engelhardt J, Storz R, Hartmann N, Bradl J and Ulrich H: Programmable beam splitter for confocal laser scanning microscopy. Progress in biomedical optics and imaging SPIE 3:13 (2002) 16–27.
- Borlinghaus R, Gugel H, Albertano P and Seyfried V: Closing the spectral gap – the transition from fixed-parameter fluorescence to tunable devices in confocal microscopy. Proc of SPIE 6090 (2006).