STELLARIS STED Stimulated Emission Depletion Microscope
Get faster to the truth
Our STimulated Emission Depletion (STED) technology joins our confocal STELLARIS platform to provide you the fastest way of imaging beyond the diffraction limit. Obtain cutting-edge nanoscopy results in no time with astounding image quality and resolution, while protecting your sample. STED super-resolution microscopy allows you to study multiple dynamic events simultaneously, so you can investigate molecular relationships and mechanisms within the cellular context.
The seamless integration of STED and our STELLARIS platform provides easy access to STED directly from the confocal interface, In a few clicks, you can get more insights from your sample, because every detail matters.
TauSTED Xtend: Sharp. Simple. Stunning.
TauSTED Xtend is the next step in the evolution of TauSTED imaging. It combines lifetime-based information with an additional spatial readout. This results in a significant gain in resolution, especially at lower STED depletion powers, allowing to extend multicolor live imaging capabilities at remarkable nanoscale. Download app note and learn more about TauSTED Xtend.
POWER
Resolve smaller details in context at nanoscale. Perform longer live imaging at cutting-edge resolution.
POTENTIAL
Use standard fluorophores and follow your preferred protocols for STED. Perform expanded multicolor imaging, even with a single STED laser line.
PRODUCTIVITY
Directly monitor fast processes with nanoscale resolution. Get instant feedback on the results while performing your experiments.


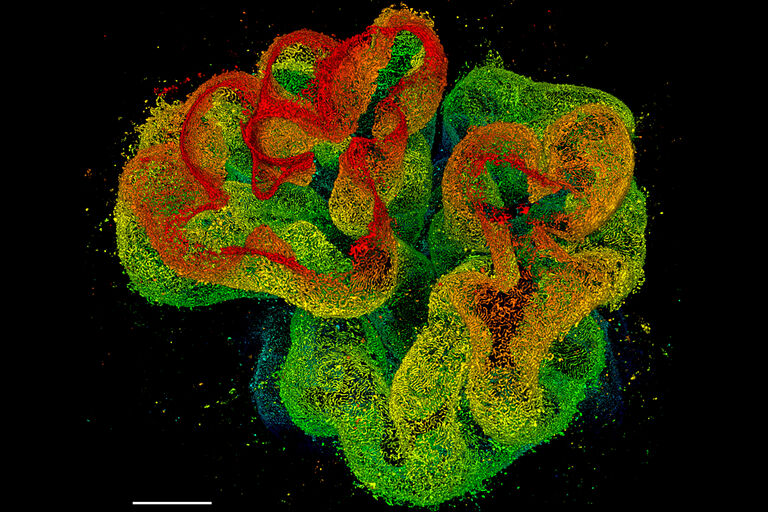
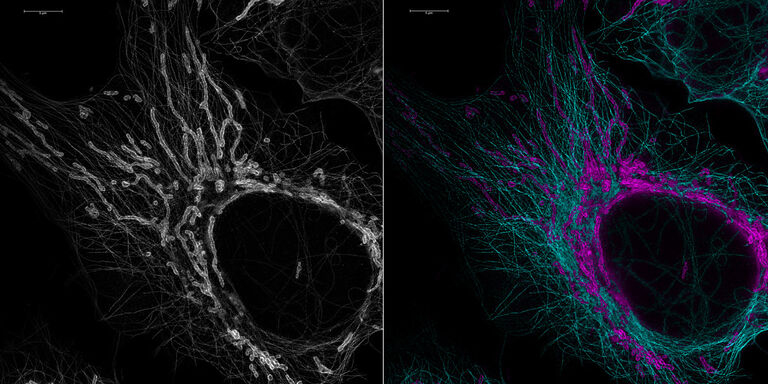
Multicolor fixed sample. TauSTED Xtend 775; Vimentin AF 594 (cyan), Phalloidin ATTO 647N (magenta) and NUP107 CF680R (glow). Image width: 39 nm. Sample courtesy of Brigitte Bergner, Mariano Gonzales Pisfil, Steffen Dietzel, Core Facility Bioimaging, Biomedical Center, Ludwig-Maximilians-University, Munich, Germany
Gentle multicolor live imaging at remarkable nanoscale
Imaging dynamics within living samples is still the most challenging experimental approach. To achieve scientific relevant results, the illumination regime needs to be, dependent on your sample and fluorophore, low enough to avoid photobleaching and phototoxicity.
With STELLARIS, including the latest generation white light lasers (WLL), Power HyD detectors and access to lifetime technologies (TauSense, FALCON), multicolour live imaging and functional imaging experiments have become routine in confocal imaging.
With TauSTED Xtend, you can significantly lower the excitation and STED light dose to such a level that allows you to study biological processes over extended periods of time even at nanoscale level.
Resolve smaller details in context at nanoscale
Are you looking for super-resolution strategies that allow you to follow your biological targets of interest in time and space with minimal external perturbations?
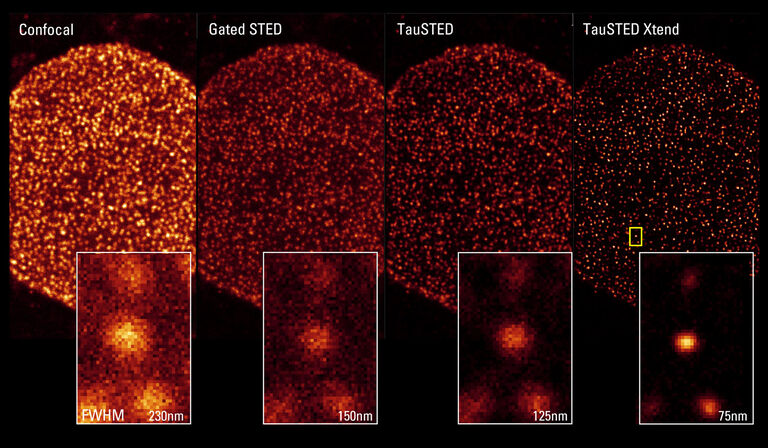
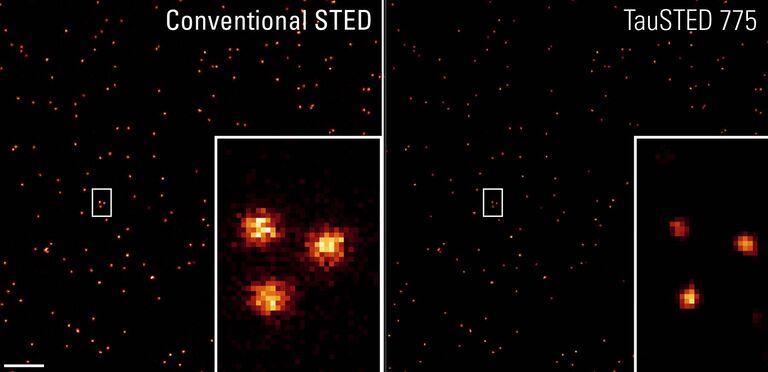
TauSTED already significantly reduces excitation and STED light doses, by significantly improving resolution versus standard confocal imaging as well as classical gated STED approaches.
TauSTED Xtend goes further by improving the maximum achievable resolution and reveals an unambiguous signal, even at lower STED depletion power.
Pushing resolution beyond conventional limits
TauSTED measures the fluorescence lifetime-based information acquired in each STED experiment and maps the STED response of the fluorophore in real time. Access to this information allows you to increase image quality (signal-to-noise), eliminate photons from the background using physical principles and push the resolution beyond the limits of intensity-based STED, no matter which excitation and STED line (589, 660, 775) you are using.
TauSTED Xtend is the next step in the evolution of STED imaging and combines the lifetime-based information with an additional spatial readout. You will see a significant gain in resolution, especially at lower STED depletion powers, allowing you to extend live imaging capabilities at remarkable nanoscale.
Use your standard protocols
Would you like to scale your experiments down to the nanoscale, using familiar protocols and a wide range of fluorescent labels and markers commonly used in life science research?
TauSTED Xtend offers new opportunities for single and multicolor experiments with green fluorescent proteins and fluorophores that are a workhorse in life science research and are currently underused in nanoscopic studies.
You can expand the palette of fluorophores, even with a single STED laser line, leveraging the unique spectral capabilities of STELLARIS.


2c TauSTED Xtend imaging of mammalian cells labeled with CF 440 - Phalloidin (glow) and immunostained against tubulin, StarGreen (cyan). Scale bar: 2 µm
Extend the number of simultaneous events you can study at the nanoscale
Multicolor fluorescence labeling has the power to reveal relationships among intracellular species, because you can image different components with molecular specificity.
STED and STELLARIS excel at multicolor applications. You can use the best dyes in the red region of the spectrum, go for fluorescent proteins in the green, or exploit novel fluorogenic probes in the orange. You can perform colocalization studies of multiple species and resolve them with sub-diffraction detail.
Our spectral detection offers you up to 5 STED-capable Power HyD detectors, so you can extend the number of molecular players to be localized in time and space. Additionally, the digital capabilities and speed of the Power HyD detectors, with 1.5 ns full-system dead time, guarantee that you get excellent image quality in terms of signal-to-noise and background, while delivering at least 10x more photon collection/pixel dwell time compared to amplified photon detectors (APDs).
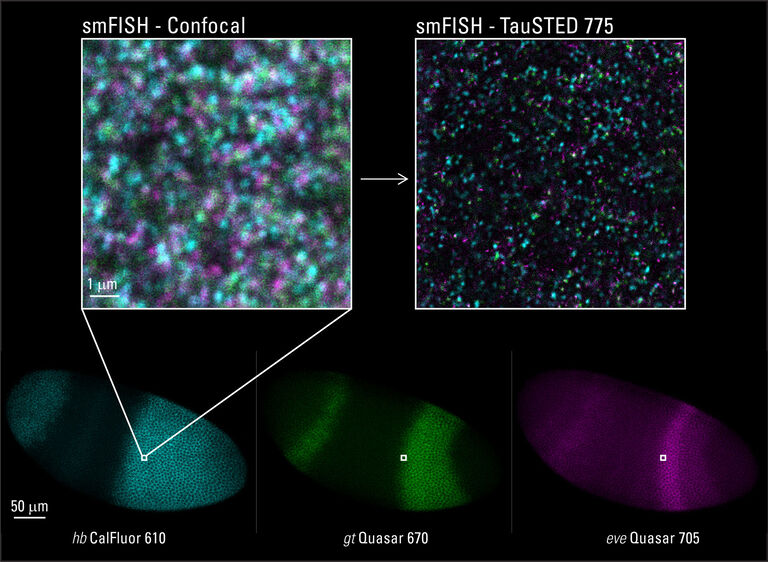
*Single molecule in-situ hybridization
Expand multicolor STED capabilities with lifetime separation
When combining STED and FALCON modalities on STELLARIS, you can expand the palette of fluorophores to study molecular interaction at nanoscopic resolution even further, as you can separate species using their fluorescence lifetime.
The emission spectra of these fluorophores cannot be distinguished using traditional intensity acquisition. Combining STED-FLIM with our automated phaser separation, the fluorophores are clearly separated using their distinct lifetimes and a single detector.
Monitor your results in time and space on-the-fly
See what happens as it happens in your experiment, in time and space with nanoscale resolution. With TauSTED Xtend, you can evaluate fast processes on the fly and receive instant feedback on the results while performing your experiments.
Ensure the highest STED performance with one-click beam auto-alignment, protecting your sample from light irradiation and achieving the optimal positioning of the laser foci in a fully automated way.
Image large areas in no time with LAS X Navigator and select your regions of interest for STED imaging.
Validate your results thanks to the capabilities of STELLARIS, such as access to raw data (ground truth), and fast and efficient photon counting.
2D and 3D STED imaging with outstanding brightness and resolution
With STELLARIS STED microscopes, you can obtain the results you need in both 2D and 3D as resolution becomes tunable in x, y and z. To ensure you obtain the best brightness and resolution in 2D and 3D, our microscopes provide you with the best optics for assuring optimal overlay of the excitation and STED PSF all over the spectrum and quantification capabilities are not lost due to adaptive corrections.
The following STED WHITE Class of objective lenses have been especially developed to guarantee optimal conditions for a variety of STED experiments:
- The STED WHITE 100x oil lens delivers the highest resolution and offers excellent performance for daily imaging of fixed samples.
- The STED WHITE 93x Glycerol lens provides adaptive correction for temperature fluctuations, refraction index mismatch, and inhomogeneities when looking deep in the sample, thanks to the motCORR fully motorized correction collar technology.
- The STED WHITE 86x Water lens with motCORR also provides adaptive correction(explained above) and is ideal for gentle live-cell applications and STED-FCS (1).
(1) “High photon count rates improve the quality of super-resolution fluorescence fluctuation spectroscopy” F. Schneider et al. J. Phys. D: Appl. Phys. 53 164003, 2020.