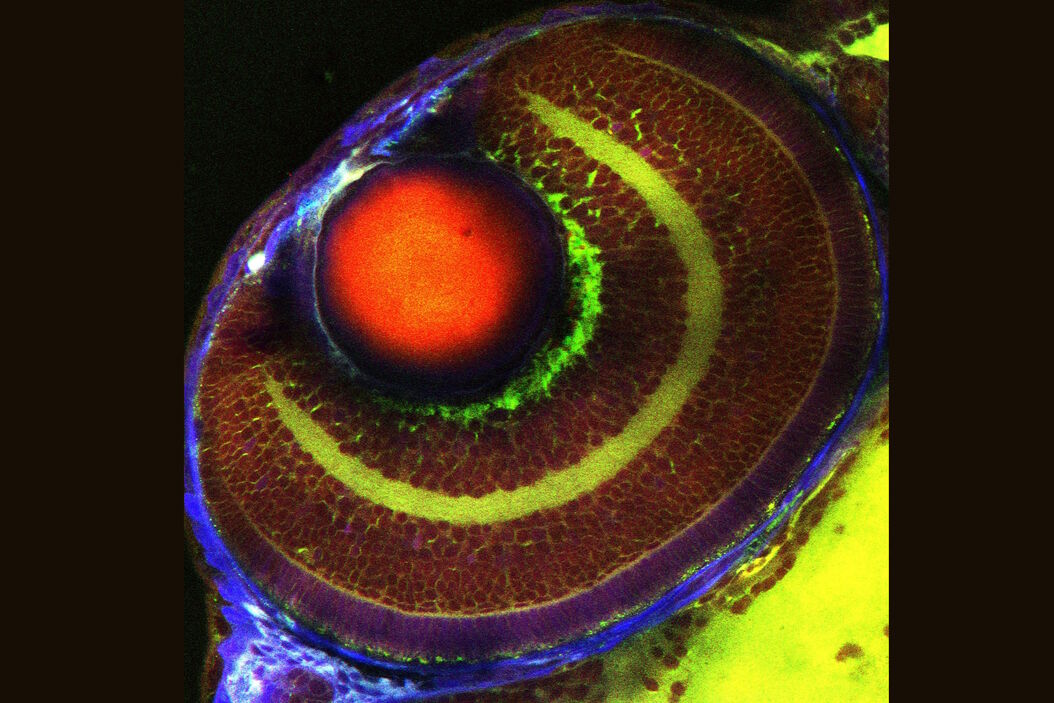
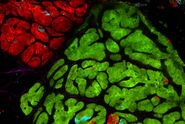
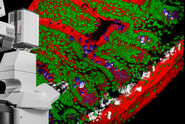
Review Articles
Illuminating life processes by vibrational probes
Qian, N., Zhao, Z., El Khoury, E. et al.
Nat Methods 22, 928–944 (2025)
Advanced vibrational microscopes for life science
Cheng, JX., Yuan, Y., Ni, H. et al.
Nat Methods 22, 912–927 (2025)
Practical considerations for quantitative and reproducible measurements with stimulated Raman scattering microscopy
Dimitrios Tsikritsis, Elizabeth J. Legge and Natalie A. Belsey
Analyst 2022,147, 4642-4656. DOI: 10.1039/d2an00817c
Emerging applications of stimulated Raman scattering microscopy in materials science
Q. Cheng,Y. Miao, J. Wild, W. Min, Y. Yang
Matter Volume 4, Issue 5, 5 May 2021, Pages 1460-1483
Biological imaging of chemical bonds by stimulated Raman scattering microscopy.
F. Hu, L. Shi, W. Min
Nat Methods. 2019 Sep;16(9):830-842. doi: 10.1038/s41592-019-0538-0. Epub 2019 Aug 30.
Cellular Imaging Using Stimulated Raman Scattering Microscopy.
A.H. Hill, D. Fu
Anal Chem. 2019 Jul 9. doi: 10.1021/acs.analchem.9b02095. [Epub ahead of print]
Vibrational spectroscopic imaging of living systems: An emerging platform for biology and medicine.
J.X. Cheng, X.S. Xie
Science. 2015 Nov 27;350(6264):aaa8870. doi: 10.1126/science.aaa8870.
Quantitative chemical imaging with stimulated Raman scattering microscopy.
D. Fu
Curr Opin Chem Biol. 2017 Aug;39:24-31. doi: 10.1016/j.cbpa.2017.05.002. Epub 2017 May 22.
Coherent Raman Scattering Microscopy in Biology and Medicine.
C. Zhang, D. Zhang, J.X. Cheng
Annu Rev Biomed Eng. 2015;17:415-45. doi: 10.1146/annurev-bioeng-071114-040554. Epub 2015 Oct 22.
Chemically sensitive bioimaging with coherent Raman scattering.
C.H. Camp, M.T. Cicerone
Nat Photonics. 2015 Apr 29;9:295-305. doi: 10.1038/nphoton.2015.60.
Applications of coherent Raman scattering microscopies to clinical and biological studies.
I.W. Schie, C. Krafft, J. Popp
Analyst. 2015 Jun 21;140(12):3897-909. doi: 10.1039/c5an00178a. Epub 2015 Mar 26.
Biological imaging with coherent Raman scattering microscopy: a tutorial.
A. Alfonso-García, R. Mittal, E.S. Lee, E.O. Potma
J Biomed Opt. 2014 Jul;19(7):71407. doi: 10.1117/1.JBO.19.7.071407.
Nonlinear vibrational microscopy applied to lipid biology.
A. Zumbusch, W. Langbein, P. Borri
Prog Lipid Res. 2013 Oct;52(4):615-32. doi: 10.1016/j.plipres.2013.07.003. Epub 2013 Sep 16.
Selected publications using Leica instruments
Multimodal imaging of a liver-on-a-chip model using labelled and label-free optical microscopy techniques
J. Maier et al,
Lab Chip, 2024,24, 4594-4608
Spectral fingerprinting of cellular lipid droplets using stimulated Raman scattering microscopy and chemometric analysis
Aurélie Rensonnet, William J. Tipping, Cedric Malherbe, Karen Faulds, Gauthier Eppe and Duncan Graham
Analyst, 2024,149, 553-562
Visualisation of drug distribution in skin using correlative optical spectroscopy and mass spectrometry imaging
N. A. Belsey et al.,
Journal of Controlled Release, 2023, 364, 79-89
A novel chalcone derivative exerts anticancer effects by promoting apoptotic cell death of human pancreatic cancer cells
Suji Baek, Sanghee Nah, Joo Yeon Park, Sang Ju Lee, Yong Gil Kang, Seung Hae Kwon, Seung Jun Oh, Kang Pa Lee, Byung Seok Moon
Bioorganic & Medicinal Chemistry 2023, 93, 117458. DOI: 10.1016/j.bmc.2023.117458
Electronically Preresonant Stimulated Raman Scattering Microscopy of Weakly Fluorescing Chromophores
Andrea Pruccoli, Mustafa Kocademir, Martin J. Winterhalder, and Andreas Zumbusch
J. Phys. Chem. B 2023, 127, 27, 6029–6037. DOI: 10.1021/acs.jpcb.3c01407
Label-Free Cytometric Evaluation of Mitosis via Stimulated Raman Scattering Microscopy and Spectral Phasor Analysis
Ewan W. Hislop, William J. Tipping, Karen Faulds, and Duncan Graham
Anal. Chem. 2023, 95, 7244−7253. DOI: 10.1021/acs.analchem.3c00212
Determination of Intracellular Esterase Activity Using Ratiometric Raman Sensing and Spectral Phasor Analysis
Henry J. Braddick, William J. Tipping, Liam T. Wilson, Harry S. Jaconelli, Emma K. Grant, Karen Faulds,* Duncan Graham,* and Nicholas C. O. Tomkinson*
Anal. Chem. 2023, 95, 5369−5376. DOI: 10.1021/acs.analchem.2c05708
Measurement of chemical penetration in skin using Stimulated Raman scattering microscopy and multivariate curve resolution - alternating least squares
Anukrati Goel, Dimitrios Tsikritsis, Natalie A. Belsey, Ruth Pendlington, Stephen Glavin, Tao Chen
Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy
Volume 296, 5 August 2023, 122639. DOI: 10.1016/j.saa.2023.122639
Spectral Unmixing for Label-Free, In-Liquid Characterization of Biomass Microstructure and Biopolymer Content by Coherent Raman Imaging
Simon Vilms Pedersen, Jonathan R. Brewer, Martin A.B. Hedegaard, and Eva Arnspang Christensen
Anal. Chem. 2023, 95, 2168−2175. DOI: 10.1021/acs.analchem.2c02349
Stimulated Raman scattering microscopy with spectral phasor analysis: applications in assessing drug–cell interactions
William J. Tipping, Liam T. Wilson, Connie An, Aristea A. Leventi, Alastair W. Wark, Corinna Wetherill, Nicholas C. O. Tomkinson, Karen Faulds and Duncan Graham
Chem. Sci., 2022, 13, 3468. DOI: 10.1039/d1sc06976d
Temporal imaging of drug dynamics in live cells using stimulated Raman scattering microscopy and a perfusion cell culture system
William J. Tipping, Andrew S. Merchant, Rebecca Fearon, Nicholas C. O. Tomkinson, Karen Faulds and Duncan Graham
RSC Chem. Biol., 2022, 3, 1154. DOI: 10.1039/d2cb00160h
Label-Free Imaging of Lipid Droplets in Prostate Cells Using Stimulated Raman Scattering Microscopy and Multivariate Analysis
Ewan W. Hislop, William J. Tipping, Karen Faulds, and Duncan Graham
Anal. Chem. 2022, 94, 8899−8908. DOI: 10.1021/acs.analchem.2c00236
Multivariate analysis of hyperspectral stimulated Raman scattering microscopy images
Imaiyan Chitra Ragupathy, Volker Schweikhard, Andreas Zumbusch
J Raman Spectrosc. 2021; 52:1630–1642. DOI: 10.1002/jrs.6184
A new class of ratiometric small molecule intracellular pH sensors for Raman microscopy
Liam T. Wilson, William J. Tipping, Lauren E. Jamieson, Corinna Wetherill, Zoë Henley, Karen Faulds, Duncan Graham, Simon P. Mackay d and Nicholas C. O. Tomkinson
Analyst, 2020, 145, 5289. DOI: 10.1039/d0an00865f
Lipid-droplet-accumulating microglia represent a dysfunctional and proinflammatory state in the aging brain
J. Marschallinger, T. Iram, M. Zardeneta, S.E. Lee, B. Lehallier, M.S. Haney, J.V. Pluvinage, V. Mathur, O. Hahn, D.W. Morgens, J. Kim, J. Tevini, T.K. Felde, H. Wolinski, C.R. Bertozzi, M.C. Bassik, L. Aigner, T. Wyss-Coray
Nat Neurosci. 2020 Feb;23(2):194-208. doi: 10.1038/s41593-019-0566-1. Epub 2020 Jan 20.
Label-free characterization of Amyloid-β-plaques and associated lipids in brain tissues using stimulated Raman scattering microscopy
V. Schweikhard, A. Baral, V. Krishnamachari, W.C. Hay, M. Fuhrmann
doi: https://doi.org/10.1101/789248
Lewy pathology in Parkinson's disease consists of crowded organelles and lipid membranes
S.H. Shahmoradian, C. Genoud, A. Graff-Meyer, J. Hench, T. Moors, G. Schweighauser, J. Wang, K.N. Goldie, R. Suetterlin, D.Castano-Diez, P. Perez-Navarro, E. Huisman, S. Ipsen, A. Ingrassia, Y. de Gier, A.J.M. Rozemuller, A. Da Paepe, J. Erny, A. Staempfli, J. Hoernschemeyer, F. Grosserueschkamp, D. Niedieker, S.F. El-Mashtoly, M. Quadri, W.F.J. van Ijcken, V. Bonifati, K. Gerwert, B. Bohrmann, S. Frank, M. Britschgi, H. Stahlberg, W. van de Berg, M.E. Lauer
Nat Neurosci. 2019 Jul;22(7):1099-1109. doi: 10.1038/s41593-019-0423-2. Epub 2019 Jun 24.
Chemical fingerprinting of single glandular trichomes of Cannabis sativa by Coherent anti-Stokes Raman scattering (CARS) microscopy,
P. Ebersbach, F. Stehle, O. Kayser, E. Freier.
BMC Plant Biology 2018 18:275 doi: https://doi.org/10.1186/s12870-018-1481-4
Cell-Nanoparticle Interactions at Sub(nanometer) Resolution Analysed by Electron Microscopy and Correlative Coherent Anti-Stokes Raman Scattering,
J. Saarinen, F. Gütter, M. Lindman, M. Agopov, S.J. Fraser-Miller, R. Scherließ, E. Jokitalo, H.A. Santos, L. Peltonen, A. Isomäki, C.J. Strachan
Biotechnol J. 2018 Oct 23. doi: 10.1002/biot.201800413.
Understanding Dissolution and Crystallization with Imaging: A Surface Point of View,
D. Novakovic, A. Isomäki, B. Pleunis, S.J. Fraser-Miller, L. Peltonen, T. Laaksonen, C.J. Strachan
Mol Pharm. 2018 Oct 9. doi: 10.1021/acs.molpharmaceut.8b00840.
Preparation and characterization of multi-component tablets containing co-amorphous salts: Combining multimodal non-linear optical imaging with established analytical methods,
R. Ojarinta, J. Saarinen, C.J. Strachan, O. Korhonen, R. Laitinen
Eur J Pharm Biopharm. 2018 Nov;132:112-126. doi: 10.1016/j.ejpb.2018.09.013. Epub 2018 Sep 22.
Chemical imaging of protein hydrogels undergoing alkaline dissolution by CARS microscopy.
B. Xu, X.D. Chen, R. Mercadé-Prieto
Food Chem. 2018 Jun 30;252:16-21. doi: 10.1016/j.foodchem.2018.01.056. Epub 2018 Jan 6.
Label-free identification of myopathological features with coherent anti-Stokes Raman scattering.
D. Niedieker, F. GrosserÜschkamp, A. Schreiner, K. Barkovits, C. Kötting, K. Marcus, K. Gerwert, M. Vorgerd
Muscle Nerve. 2018 Apr 16. doi: 10.1002/mus.26140. [Epub ahead of print]
Variability in responses observed in human white adipose tissue models.
R.D. Abbott, F.E. Borowsky, C.A. Alonzo, A. Zieba, I. Georgakoudi, D.L. Kaplan
J Tissue Eng Regen Med. 2018 Mar;12(3):840-847. doi: 10.1002/term.2572. Epub 2017 Nov 10.
Ostreococcus tauri is a high-lipid content green algae that extrudes clustered lipid droplets
C.R. Smallwood, W. Chrisler, J.H. Chen, E. Patello, M. Thomas, R. Boudreau, A. Ekman, H. Wang, G. McDermott, J.E. Evans
doi: 10.1101/249052
Chemical basis for alteration of an intraocular lens using a femtosecond laser.
J.F. Bille et al.
Biomed. Opt. Express 8, 1390-1404, (2017)
Noninvasive Diagnosis of High-Grade Urothelial Carcinoma in Urine by Raman Spectral Imaging.
H.K. Yosef, S.D. Krauß, T. Lechtonen, H. Jütte, A. Tannapfel, H.U. Käfferlein, T. Brüning, F. Roghmann, J. Noldus, A. Mosig, S.F. El-Mashtoly, K. Gerwert
Anal Chem. 2017 Jun 20;89(12):6893-6899. doi: 10.1021/acs.analchem.7b01403. Epub 2017 Jun 5.
Role for formin-like 1-dependent acto-myosin assembly in lipid droplet dynamics and lipid storage.
S.G. Pfisterer, G. Gateva, P. Horvath, J. Pirhonen, V.T. Salo, L. Karhinen, M. Varjosalo, S.J. Ryhänen, P. Lappalainen, E. Ikonen
Nat Commun. 2017 Mar 31;8:14858. doi: 10.1038/ncomms14858.
Insights into Caco-2 cell culture structure using coherent anti-Stokes Raman scattering (CARS) microscopy.
J. Saarinen, E. Sözeri, S.J. Fraser-Miller, L Peltonen, H.A. Santos, A. Isomäki, C.J. Strachan
Int J Pharm. 2017 May 15;523(1):270-280. doi: 10.1016/j.ijpharm.2017.03.015. Epub 2017 Mar 12.
Noninvasive Diagnosis of High-Grade Urothelial Carcinoma in Urine by Raman Spectral Imaging.
H.K. Yosef, S.D. Krauß, T. Lechtonen, H. Jütte, A. Tannapfel, H.U. Käfferlein, T. Brüning, F. Roghmann, J. Noldus, A. Mosig, S.F. El-Mashtoly, K. Gerwert
Anal Chem. 2017 Jun 20;89(12):6893-6899. doi: 10.1021/acs.analchem.7b01403. Epub 2017 Jun 5.
Virtual staining of colon cancer tissue by label-free Raman micro-spectroscopy.
D. Petersen, L. Mavarani, D. Niedieker, E. Freier, A. Tannapfel, C. Kötting, K. Gerwert, S.F. El-Mashtoly
Analyst. 2017 Apr 10;142(8):1207-1215. doi: 10.1039/c6an02072k.
Coupling of vinculin to F-actin demands Syndecan-4 proteoglycan.
R.P. Cavalheiro, M.A. Lima, T.R. Jarrouge-Bouças, G.M. Viana, C.C. Lopes, V.J. Coulson-Thomas, J.L. Dreyfuss, E.A. Yates, I.L.S. Tersariol, H.B. Nader
Matrix Biol. 2017 Nov;63:23-37. doi: 10.1016/j.matbio.2016.12.006. Epub 2017 Jan 4.
Direct visualization of the arterial wall water permeability barrier using CARS microscopy.
B.M. Lucotte, C. Powell, J.R. Knutson, C.A. Combs, D. Malide, Z.X. Yu, M. Knepper, K.D. Patel, A. Pielach, E. Johnson, L. Borysova, K.A. Dora, R.S. Balaban
Proc Natl Acad Sci U S A. 2017 May 2;114(18):4805-4810. doi: 10.1073/pnas.1620008114. Epub 2017 Apr 3.
Watching dance of the molecules - CARS microscopy.
[Article in Polish; Abstract available in Polish from the publisher]
J. Korczyński, K. Kubiak, E. Węgłowska
Postepy Biochem. 2017;63(1):44-52.
Label-free in situ imaging of oil body dynamics and chemistry in germination
G.Waschatko, N. Billecke, S. Schwendy, H. Jaurich, M. Bonn, T.A. Vilgis and S.H. Parekh
J R Soc Interface. 2016 Oct; 13(123): 20160677. doi: 10.1098/rsif.2016.0677
Continuous grading of early fibrosis in NAFLD using label-free imaging: A proof-of-concept study.
Pirhonen, J. et al.
PloS one 11, e0147804, (2016).
Raman spectroscopy and coherent anti-Stokes Raman scattering imaging: prospective tools for monitoring skeletal cells and skeletal regeneration.
C. C. Moura, R. S. Tare, R. O. C. Oreffo & S. Mahajan
J R Soc Interface 13, 20160182-, (2016).
Elucidation of Compression-Induced Surface Crystallization in Amorphous Tablets Using Sum Frequency Generation (SFG) Microscopy.
Mah, P. T. et al.
Pharmaceutical Research, 1-14, (2016).
19F labelled glycosaminoglycan probes for solution NMR and non-linear (CARS) microscopy.
Lima, M. A. et al.
Glycoconjugate Journal, 1-6, (2016).
A novel multi-modal platform to image molecular and elemental alterations in ischemic stroke.
Caine, S. et al.
Neurobiology of Disease 91, 132-142, (2016).
Label-free visualization of cholesteatoma in the mastoid and tympanic membrane using CARS microscopy
J. Zou, A. Isomäki, T. Hirvonen, A. Aarnisalo, J. Jero, I. Pyykkö
Journal of Otology 2016; 11(3): 127. doi:10.1016/j.joto.2016.09.001
Multimodal non-linear optical imaging for the investigation of drug nano-/microcrystal-cell interactions.
Darville, N. et al.
Eur J Pharm Biopharm 96, 338-348, (2015).
Biochemical and Bioimaging Evidence of Cholesterol in Acquired Cholesteatoma.
B. Thorsted, M. Bloksgaard, A. Groza, L.P. Schousboe, N.J. Færgeman, J.A. Sørensen, V. Svane-Knudsen, J.R. Brewer
Ann Otol Rhinol Laryngol. 2016 Aug;125(8):627-33. doi: 10.1177/0003489416642784. Epub 2016 Apr 15.
Lipid droplets as a novel cargo of tunnelling nanotubes in endothelial cells
K. Astanina, M. Koch, C. Jüngst, A. Zumbusch and A.K. Kiemer
Sci Rep. 2015; 5: 11453. Published online 2015 Jun 22. doi: 10.1038/srep11453
Fully automated registration of vibrational microspectroscopic images in histologically stained tissue sections.
C. Yang, D. Niedieker, F. Grosserüschkamp, M. Horn, A. Tannapfel, A. Kallenbach-Thieltges, K. Gerwert, A. Mosig
BMC Bioinformatics. 2015 Nov 25;16:396. doi: 10.1186/s12859-015-0804-9.
Colocalization of fluorescence and Raman microscopic images for the identification of subcellular compartments: a validation study.
S.D. Krauß, D. Petersen, D. Niedieker, I. Fricke, E. Freier, S.F. El-Mashtoly, K. Gerwert, A. Mosig
Analyst. 2015 Apr 7;140(7):2360-8. doi: 10.1039/c4an02153c.
Perilipin-related protein regulates lipid metabolism in C. elegans.
A.A. Chughtai, F. Kaššák, M. Kostrouchová, J.P. Novotný, M.W. Krause, V. Saudek, Z. Kostrouch, M. Kostrouchová
PeerJ. 2015 Sep 1;3:e1213. doi: 10.7717/peerj.1213. eCollection 2015.
Investigation of protein distribution in solid lipid particles and its impact on protein release using coherent anti-Stokes Raman scattering microscopy.
P.C. Christophersen, D. Birch, J. Saarinen, A. Isomäki, H.M. Nielsen, M. Yang, C.J. Strachan, H. Mu
J Control Release. 2015 Jan 10;197:111-20. doi: 10.1016/j.jconrel.2014.10.023. Epub 2014 Nov 3.
Adipose triglyceride lipase is involved in the mobilization of triglyceride and retinoid stores of hepatic stellate cells.
U. Taschler, R. Schreiber, C. Chitraju, G.F. Grabner, M. Romauch, H. Wolinski, G. Haemmerle, R. Breinbauer, R. Zechner, A. Lass, R. Zimmermann
Biochim Biophys Acta. 2015 Jul;1851(7):937-45. doi: 10.1016/j.bbalip.2015.02.017. Epub 2015 Feb 27.
Seipin is involved in the regulation of phosphatidic acid metabolism at a subdomain of the nuclear envelope in yeast.
H. Wolinski, H.F. Hofbauer, K. Hellauer, A. Cristobal-Sarramian, D. Kolb, M. Radulovic, O.L. Knittelfelder, G.N. Rechberger, S.D. Kohlwein
Biochim Biophys Acta. 2015 Nov;1851(11):1450-64. doi: 10.1016/j.bbalip.2015.08.003. Epub 2015 Aug 12.
Microscopic and spectroscopic techniques to investigate lipid droplet formation and turnover in yeast.
H. Wolinski, S.D. Kohlwein
Methods Mol Biol. 2015;1270:289-305. doi: 10.1007/978-1-4939-2309-0_21.
Study of carbamate-modified disiloxane in porous PVDF-HFP membranes: new electrolytes/separators for lithium-ion batteries.
Jeschke, S., Mutke, M., Jiang, Z., Alt, B. & Wiemhofer, H. D.
Chemphyschem 15, 1761-1771, (2014).
Automated identification of subcellular organelles by coherent anti-stokes Raman scattering.
S.F. El-Mashtoly, D. Niedieker, D. Petersen, S.D. Krauss, E. Freier, A. Maghnouj, A. Mosig, S. Hahn, C. Kötting, K. Gerwert
Biophys J. 2014 May 6;106(9):1910-20. doi: 10.1016/j.bpj.2014.03.025.
Lipid droplet autophagy in the yeast Saccharomyces cerevisiae.
T. van Zutphen, V. Todde,R. de Boer, M. Kreim, H.F. Hofbauer, H. Wolinski, M. Veenhuis, I.J. van der Klei, S.D. Kohlwein
Mol Biol Cell. 2014 Jan;25(2):290-301. doi: 10.1091/mbc.E13-08-0448. Epub 2013 Nov 20.
The emergence of lipid droplets in yeast: current status and experimental approaches.
M. Radulovic, O. Knittelfelder, A. Cristobal-Sarramian, D. Kolb, H. Wolinski, S.D. Kohlwein
Curr Genet. 2013 Nov;59(4):231-42. doi: 10.1007/s00294-013-0407-9. Epub 2013 Sep 21.
Remodeling of lipid droplets during lipolysis and growth in adipocytes.
M. Paar, C. Jüngst, N.A. Steiner, C. Magnes, F. Sinner, D. Kolb, A. Lass, R. Zimmermann, A. Zumbusch, S.D. Kohlwein, H. Wolinski
J Biol Chem. 2012 Mar 30;287(14):11164-73. doi: 10.1074/jbc.M111.316794. Epub 2012 Feb 6.
Quantitative imaging of lipid metabolism in yeast: from 4D analysis to high content screens of mutant libraries.
H. Wolinski, K. Bredies, S.D. Kohlwein
Methods Cell Biol. 2012;108:345-65. doi: 10.1016/B978-0-12-386487-1.00016-X.
Quantitative assessment of collagen fibre orientations from two-dimensional images of soft biological tissues.
A.J. Schriefl, A.J. Reinisch, S. Sankaran, D.M. Pierce, G.A. Holzapfel
J R Soc Interface. 2012 Nov 7;9(76):3081-93. Epub 2012 Jul 4.
Cancer Research
Profiling single cancer cell metabolism via high-content SRS imaging with chemical sparsity
Y. Tan, H. Lin, J.-X. Cheng
Science Advances (2023) 9, eadg6061. DOI: 10.1126/sciadv.adg6061
Raman-guided subcellular pharmaco-metabolomics for metastatic melanoma cells
J. Du, Y. Su, C. Qian, D. Yuan, K. Miao, D. Lee, A.H.C. Ng, R.S. Wijker, A. Ribas, R.D. Levine, J.R. Heath, L. Wei
Nat Commun. 2020 Sep 24;11(1):4830. doi: 10.1038/s41467-020-18376-x.
Virtual staining of colon cancer tissue by label-free Raman micro-spectroscopy.
D. Petersen, L. Mavarani, D. Niedieker, E. Freier, A. Tannapfel, C. Kötting, K. Gerwert, S.F. El-Mashtoly
Analyst. 2017 Apr 10;142(8):1207-1215. doi: 10.1039/c6an02072k.
Detection of human brain tumor infiltration with quantitative stimulated Raman scattering microscopy.
M. Ji, S. Lewis, S. Camelo-Piragua, S.H. Ramkissoon, M. Snuderl, S. Venneti, A. Fisher-Hubbard, M. Garrard, D. Fu, A.C. Wang, J.A. Heth, C.O. Mahe, N. Sanai, T.D. Johnson, C.W. Freudiger, O. Sagher, X.S. Xie, D.A. Orringer
Sci Transl Med. 2015 Oct 14;7(309):309ra163. doi: 10.1126/scitranslmed.aab0195.
Rapid histology of laryngeal squamous cell carcinoma with deep-learning based stimulated Raman scattering microscopy.
L. Zhang, Y. Wu, B. Zheng, L. Su, Y. Chen, S. Ma, Q. Hu, X. Zou, L. Yao, Y. Yang, L. Chen, Y. Mao, Y. Chen, M. Ji
Theranostics 2019, Vol. 9, Issue 9
Lipid Desaturation Is a Metabolic Marker and Therapeutic Target of Ovarian Cancer Stem Cells.
J. Li, S. Condello, J. Thomes-Pepin, X. Ma, Y. Xia, T.D. Hurley, D. Matei, J.X. Cheng
Cell Stem Cell. 2017 Mar 2;20(3):303-314.e5. doi: 10.1016/j.stem.2016.11.004. Epub 2016 Dec 29.
Cholesterol esterification inhibition and imatinib treatment synergistically inhibit growth of BCR-ABL mutation-independent resistant chronic myelogenous leukemia.
S. Bandyopadhyay, J. Li, E. Traer, J.W. Tyner, A. Zhou, S.T. Oh, J.X. Cheng
PLoS One. 2017 Jul 18;12(7):e0179558. doi: 10.1371/journal.pone.0179558. eCollection 2017.
Monitoring neoadjuvant therapy responses in rectal cancer using multimodal nonlinear optical microscopy.
Lian-Huang Li,Zhi-Fen Chen, Xing-Fu Wang, Xing Liu, Wei-Zhong Jiang, Shuang-Mu Zhuo, Li-Wei Jiang, Guo-Xian Guan, and Jian-Xin Chen
Oncotarget. 2017 Dec 5; 8(63): 107323–107333. Published online 2017 Nov 3. doi: 10.18632/oncotarget.22366
Cholesteryl ester accumulation induced by PTEN loss and PI3K/AKT activation underlies human prostate cancer aggressiveness.
S.Yue, J. Li, S.Y. Lee, H.J. Lee, T. Shao, B. Song, L. Cheng, T.A. Masterson, X. Liu, T.L. Ratliff, J.X. Cheng
Cell Metab. 2014 Mar 4;19(3):393-406. doi: 10.1016/j.cmet.2014.01.019.
Visualization of Breast Cancer Metabolism Using Multimodal Nonlinear Optical Microscopy of Cellular Lipids and Redox State.
J. Hou, J. Williams, E.L. Botvinick, E.O. Potma, B.J. Tromberg
Cancer Res. 2018 May 15;78(10):2503-2512. doi: 10.1158/0008-5472.CAN-17-2618. Epub 2018 Mar 13.
Multimodal Imaging/Histopathology
Hierarchical deep convolutional neural networks combine spectral and spatial information for highly accurate Raman-microscopy-based cytopathology.
S.D. Krauß, R. Roy, H.K. Yosef, T. Lechtonen, S.F. El-Mashtoly, K. Gerwert, A. Mosig
J Biophotonics. 2018 Oct;11(10):e201800022. doi: 10.1002/jbio.201800022. Epub 2018 Jul 5.
Histological coherent Raman imaging: a prognostic review.
M.T. Cicerone, C.H. Camp
Analyst. 2017 Dec 18;143(1):33-59. doi: 10.1039/c7an01266g.
Detection of human brain tumor infiltration with quantitative stimulated Raman scattering microscopy.
M. Ji, S. Lewis, S. Camelo-Piragua, S.H. Ramkissoon, M. Snuderl, S. Venneti, A. Fisher-Hubbard, M. Garrard, D. Fu, A.C. Wang, J.A. Heth, C.O. Maher, N. Sanai, T.D. Johnson, C.W. Freudiger, O. Sagher, X.S. Xie, D.A. Orringer.
Sci Transl Med. 2015 Oct 14;7(309):309ra163. doi: 10.1126/scitranslmed.aab0195.
Fluorescence lifetime imaging of endogenous biomarker of oxidative stress.
R. Datta, A. Alfonso-García, R. Cinco, E. Gratton
Sci Rep. 2015 May 20;5:9848. doi: 10.1038/srep09848.
Label-free multiphoton microscopy reveals altered tissue architecture in hippocampal sclerosis.
O. Uckermann, R. Galli, S. Leupold, R. Coras, M. Meinhardt, S. Hallmeyer-Elgner, T. Mayer, A. Storch, G. Schackert, E. Koch, I. Blümcke, G. Steiner, M. Kirsch
Epilepsia. 2017 Jan;58(1):e1-e5. doi: 10.1111/epi.13598. Epub 2016 Nov 7.
Pseudo-HE images derived from CARS/TPEF/SHG multimodal imaging in combination with Raman-spectroscopy as a pathological screening tool.
T.W. Bocklitz, F.S. Salah, N. Vogler, S. Heuke, O. Chernavskaia, C. Schmidt, M.J. Waldner, F.R. Greten, R. Bräuer, M. Schmitt, A. Stallmach, I. Petersen, J. Popp
BMC Cancer. 2016 Jul 26;16:534. doi: 10.1186/s12885-016-2520-x.
Multimodal nonlinear microscopy of head and neck carcinoma - toward surgery assisting frozen section analysis.
S. Heuke, O. Chernavskaia, T. Bocklitz, F.B. Legesse, T. Meyer, D. Akimov, O. Dirsch, G. Ernst, F. von Eggeling, I. Petersen, O. Guntinas-Lichius, M. Schmitt, J. Popp
Head Neck. 2016 Oct;38(10):1545-52. doi: 10.1002/hed.24477. Epub 2016 Apr 21.
A quantitative framework for the analysis of multimodal optical microscopy images
A.J. Bower, B. Chidester, J. Li, Y. Zhao, M. Marjanovic, E.J. Chaney, M.N. Do and S.A. Boppart
Quant Imaging Med Surg. 2017 Feb; 7(1): 24–37. doi: 10.21037/qims.2017.02.07
High-Speed Coherent Raman Fingerprint Imaging of Biological Tissues.
C.H. Camp Jr, Y.J. Lee, J.M. Heddleston, C.M. Hartshorn, A.R. Hight Walker, J.N. Rich, J.D. Lathia, M.T. Cicerone.
Nat Photonics. 2014;8:627-634.
Neuroscience
Lipid-droplet-accumulating microglia represent a dysfunctional and proinflammatory state in the aging brain
J. Marschallinger, T. Iram, M. Zardeneta, S.E. Lee, B. Lehallier, M.S. Haney, J.V. Pluvinage, V. Mathur, O. Hahn, D.W. Morgens, J. Kim, J. Tevini, T.K. Felde, H. Wolinski, C.R. Bertozzi, M.C. Bassik, L. Aigner, T. Wyss-Coray
Nat Neurosci. 2020 Feb;23(2):194-208. doi: 10.1038/s41593-019-0566-1. Epub 2020 Jan 20.
Lewy pathology in Parkinson's disease consists of crowded organelles and lipid membranes
S.H. Shahmoradian, C. Genoud, A. Graff-Meyer, J. Hench, T. Moors, G. Schweighauser, J. Wang, K.N. Goldie, R. Suetterlin, D.Castano-Diez, P. Perez-Navarro, E. Huisman, S. Ipsen, A. Ingrassia, Y. de Gier, A.J.M. Rozemuller, A. Da Paepe, J. Erny, A. Staempfli, J. Hoernschemeyer, F. Grosserueschkamp, D. Niedieker, S.F. El-Mashtoly, M. Quadri, W.F.J. van Ijcken, V. Bonifati, K. Gerwert, B. Bohrmann, S. Frank, M. Britschgi, H. Stahlberg, W. van de Berg, M.E. Lauer
Nat Neurosci. 2019 Jul;22(7):1099-1109. doi: 10.1038/s41593-019-0423-2. Epub 2019 Jun 24.
Label-free imaging of amyloid plaques in Alzheimer’s disease with stimulated Raman scattering microscopy,
M. Ji, M.Arbel, L. Zhang, C.W. Freudiger, S.S. Hou, D. Lin, X. Yang, B.J. Bacskai and X.S. Xie.
Sci Adv. 2018 Nov; 4(11): eaat7715. Published online 2018 Nov 16. doi: 10.1126/sciadv.aat7715
Label-Free Vibrational Spectroscopic Imaging of Neuronal Membrane Potential.
H.J. Lee, D. Zhang, Y. Jiang, X. Wu, P.Y. Shih, C.S. Liao, B. Bungart, X.M. Xu, R. Drenan, E. Bartlett, J.X. Cheng
J Phys Chem Lett. 2017 May 4;8(9):1932-1936. doi: 10.1021/acs.jpclett.7b00575. Epub 2017 Apr 17.
Label-free Imaging of Neurotransmitter Acetylcholine at Neuromuscular Junctions with Stimulated Raman Scattering.
D. Fu, W. Yang, X.S. Xie
J Am Chem Soc. 2017 Jan 18;139(2):583-586. doi: 10.1021/jacs.6b10727. Epub 2016 Dec 30.
Bioorthogonal chemical imaging of metabolic activities in live mammalian hippocampal tissues with stimulated Raman scattering.
F. Hu, M.R. Lamprecht, L. Wei, B. Morrison, W. Min
Sci Rep. 2016 Dec 21;6:39660. doi: 10.1038/srep39660.
Monitoring peripheral nerve degeneration in ALS by label-free stimulated Raman scattering imaging.
F. Tian, W. Yang, D.A. Mordes, J.Y. Wang, J.S. Salameh, J. Mok, J. Chew, A. Sharma, E. Leno-Duran, S. Suzuki-Uematsu, N. Suzuki, S.S. Han, F.K. Lu, M. Ji, R. Zhang, Y. Liu, J. Strominger, N.A. Shneider, L. Petrucelli, X.S. Xie, K. Eggan
Nat Commun. 2016 Oct 31;7:13283. doi: 10.1038/ncomms13283.
Probing pain pathways with light.
F. Wang, E. Bélanger, M.E. Paquet, D.C. Côté, Y. De Koninck
Neuroscience. 2016 Dec 3;338:248-271. doi: 10.1016/j.neuroscience.2016.09.035. Epub 2016 Oct 1.
Plaque-associated lipids in Alzheimer’s diseased brain tissue visualized by nonlinear microscopy
J. Kiskis, H. Fink, L. Nyberg, J. Thyr, J.Y. Li and A. Enejder
Sci Rep. 2015; 5: 13489. Published online 2015 Aug 27. doi: 10.1038/srep13489
Pharmacology
Raman Imaging of Nanocarriers for Drug Delivery.
S. Vanden-Hehir, W.J. Tipping, M. Lee, V.G. Brunton, A. Williams, A.N. Hulme
Nanomaterials (Basel). 2019 Mar 3;9(3). pii: E341. doi: 10.3390/nano9030341.
Detecting and Quantifying Microscale Chemical Reactions in Pharmaceutical Tablets by Stimulated Raman Scattering Microscopy.
B. Figueroa, T. Nguyen, S. Sotthivirat, W. Xu, T. Rhodes, M.S. Lamm, R.L. Smith, C.T. John, Y. Su, D. Fu
Anal Chem. 2019 May 21;91(10):6894-6901. doi: 10.1021/acs.analchem.9b01269. Epub 2019 Apr 30.
In Situ Stimulated Raman Scattering (SRS) Microscopy Study of the Dissolution of Sustained-Release Implant Formulation.
A.T. Francis, T.T. Nguyen, M.S. Lamm, R. Teller, S.P. Forster, W. Xu, T. Rhodes, R.L. Smith, J. Kuiper, Y. Su, D. Fu
Mol Pharm. 2018 Dec 3;15(12):5793-5801. doi: 10.1021/acs.molpharmaceut.8b00965. Epub 2018 Nov 8.
Imaging the Intracellular Distribution of Tyrosine Kinase Inhibitors in Living Cells with Quantitative Hyperspectral Stimulated Raman Scattering
D. Fu, J. Zhou, W.S. Zhu, P.W. Manley,Y.K. Wang, T. Hood, A. Wylie, X.S. Xie
Nat Chem. 2014 Jul; 6(7): 614–622. doi: 10.1038/nchem.1961
Stimulated Raman scattering microscopy: an emerging tool for drug discovery.
W.J. Tipping, M. Lee, A. Serrels, V.G. Brunton, A.N. Hulme
Chem Soc Rev. 2016 Apr 21;45(8):2075-89. doi: 10.1039/c5cs00693g. Epub 2016 Feb 3.
Determination of the Subcellular Localization and Mechanism of Action of Ferrostatins in Suppressing Ferroptosis.
M.M. Gaschler, F. Hu, H. Feng, A. Linkermann, W. Min, B.R. Stockwell
ACS Chem Biol. 2018 Apr 20;13(4):1013-1020. doi: 10.1021/acschembio.8b00199. Epub 2018 Mar 13.
Multimodal Nonlinear Optical Imaging for Sensitive Detection of Multiple Pharmaceutical Solid-State Forms and Surface Transformations.
D. Novakovic, J. Saarinen, T. Rojalin, O. Antikainen, S.J. Fraser-Miller, T. Laaksonen, L. Peltonen, A. Isomäki, C.J. Strachan
Anal Chem. 2017 Nov 7;89(21):11460-11467. doi: 10.1021/acs.analchem.7b02639. Epub 2017 Oct 18.
Intracellular imaging of docosanol in living cells by coherent anti-Stokes Raman scattering microscopy.
S. You, Y. Liu, Z. Arp, Y. Zhao, E.J. Chaney, M. Marjanovic, S.A. Boppart
J Biomed Opt. 2017 Jul 1;22(7):70502. doi: 10.1117/1.JBO.22.7.070502.
Metabolism
Spectral tracing of deuterium for imaging glucose metabolism
L. Zhang, L. Shi, Y. Shen, Y. Miao, M. Wei, N. Qian, Y. Liu, and W. Min
Nat Biomed Eng. 2019 May; 3(5): 402–413. Published online 2019 Apr 29. doi: 10.1038/s41551-019-0393-4
Optical imaging of metabolic dynamics in animals
L. Shi, C.Zheng, Y. Shen, Z. Chen, E.S. Silveira, L. Zhang, M. Wei, C. Liu, C. de Sena-Tomas, K. Targoff and W. Min
Nat Commun. 2018; 9: 2995. Published online 2018 Aug 6. doi: 10.1038/s41467-018-05401-3
Multicolor Activatable Raman Probes for Simultaneous Detection of Plural Enzyme Activities
H. Fujioka, J. Shou, R. Kojima, Y. Urano, Y. Ozeki and M. Kamiya
J. Am. Chem. Soc. 2020, 142, 49, 20701–20707, Publication Date: November 23, 2020.
Metabolic activity induces membrane phase separation in endoplasmic reticulum.
Shen Y, Zhao Z, Zhang L, Shi L, Shahriar S, Chan RB, Di Paolo G and Min W.
PNAS 2017, 114(51):13394-13399. DOI: 10.1073/pnas.1712555114
Two-color vibrational imaging of glucose metabolism using stimulated Raman scattering.
Long R, Zhang L, Shi L, Shen Y, Hu F, Zeng C, Min W.
Chem Commun (Camb). 2018 Jan 7;54(2):152-155. doi: 10.1039/c7cc08217g. Epub 2017 Dec 8.
Microscopy tools for the investigation of intracellular lipid storage and dynamics.
Daemen S, van Zandvoort MAMJ, Parekh SH, Hesselink MKC.
Mol Metab. 2015 Dec 31;5(3):153-163. doi: 10.1016/j.molmet.2015.12.005. eCollection 2016 Mar.
Imaging complex protein metabolism in live organisms by stimulated Raman scattering microscopy with isotope labeling.
Wei L, Shen Y, Xu F, Hu F, Harrington JK, Targoff KL, Min W.
ACS Chem Biol. 2015 Mar 20;10(3):901-8. doi: 10.1021/cb500787b. Epub 2015 Jan 15.
Assessing cholesterol storage in live cells and C. elegans by stimulated Raman scattering imaging of phenyl-Diyne cholesterol.
Lee HJ, Zhang W, Zhang D, Yang Y, Liu B, Barker EL, Buhman KK, Slipchenko LV, Dai M, Cheng JX.
Sci Rep. 2015 Jan 22;5:7930. doi: 10.1038/srep07930.
Label-free DNA imaging in vivo with stimulated Raman scattering microscopy.
Lu FK, Basu S, Igras V, Hoang MP, Ji M, Fu D, Holtom GR, Neel VA, Freudiger CW, Fisher DE, Xie XS.
Proc Natl Acad Sci U S A. 2015 Sep 15;112(37):11624-9. doi: 10.1073/pnas.1515121112. Epub 2015 Aug 31.
Immunology
Beyond endoscopic assessment in inflammatory bowel disease: real-time histology of disease activity by non-linear multimodal imaging.
O. Chernavskaia, S. Heuke, M. Vieth, O. Friedrich, S. Schürmann, R. Atreya, A. Stallmach, M.F. Neurath, M. Waldner, I. Petersen, M. Schmitt, T. Bocklitz, J. Popp
Sci Rep. 2016 Jul 13;6:29239. doi: 10.1038/srep29239.
Lipid biochemical changes detected in normal appearing white matter of chronic multiple sclerosis by spectral coherent Raman imaging
K.W.C. Poon, C. Brideau, R. Klaver, G. J. Schenk, J.J. Geurts and P.K. Stys
Chem Sci. 2018 Feb 14; 9(6): 1586–1595. Published online 2018 Jan 2. doi: 10.1039/c7sc03992a
Plant Research/Biofuels
In situ chemically specific mapping of agrochemical seed coatings using stimulated Raman scattering microscopy.
C.C. Wang, S. Moorhouse, C. Stain, M. Seymour, E. Green, S. Penfield, J. Moger
J Biophotonics. 2018 May 17:e201800108. doi: 10.1002/jbio.201800108. [Epub ahead of print]
Visualizing chemical functionality in plant cell walls.
Y. Zeng, M.E. Himmel, S.Y. Ding
Biotechnol Biofuels. 2017 Nov 30;10:263. doi: 10.1186/s13068-017-0953-3. eCollection 2017.
Probing the metabolic heterogeneity of live Euglena gracilis with stimulated Raman scattering microscopy.
Y. Wakisaka, Y. Suzuki, O. Iwata, A. Nakashima, T. Ito, M. Hirose, R. Domon, M. Sugawara, N. Tsumura, H. Watarai, T. Shimobaba, K. Suzuki, K. Goda, Y. Ozeki
Nat Microbiol. 2016 Aug 1;1(10):16124. doi: 10.1038/nmicrobiol.2016.124.
Materials Science
Emerging applications of stimulated Raman scattering microscopy in materials science
Q. Cheng,Y. Miao, J. Wild, W. Min, Y. Yang
Matter Volume 4, Issue 5, 5 May 2021, Pages 1460-1483
Operando and three-dimensional visualization of anion depletion and lithium growth by stimulated Raman scattering microscopy
Q. Cheng, L. Wei, Z. Liu, N. Ni, Z. Sang, B. Zhu, W. Xu, M. Chen, Y. Miao, L.Q. Chen, W. Min, Y. Yang
Nature Communications volume 9, Article number: 2942 (2018)
Quantitative Chemical Imaging of Nonplanar Microfluidics.
M.K. Gelber, M.R. Kole, N. Kim, N.R. Aluru, R. Bhargava
Anal Chem. 2017 Feb 7;89(3):1716-1723. doi: 10.1021/acs.analchem.6b03943. Epub 2017 Jan 9.
Coherent anti-Stokes Raman scattering microscopy of single nanodiamonds
I. Pope, L. Payne, G. Zoriniants, E. Thomas, O. Williams, P. Watson, W. Langbein and P. Borri
Nat Nanotechnol. 2014 Nov; 9(11): 940–946. doi: 10.1038/nnano.2014.210
Vibrational Tags
Raman Imaging of Small Biomolecules
Y. Shen, F. Hu, W. Min
Annu Rev Biophys. 2019 May 6;48:347-369. doi: 10.1146/annurev-biophys-052118-115500. Epub 2019 Mar 20.
Spectral tracing of deuterium for imaging glucose metabolism
L. Zhang, L. Shi, Y. Shen, Y. Miao, M. Wei, N. Qian, Y. Liu, and W. Min
Nat Biomed Eng. 2019 May; 3(5): 402–413. Published online 2019 Apr 29. doi: 10.1038/s41551-019-0393-4
Optical imaging of metabolic dynamics in animals
L. Shi, C.Zheng, Y. Shen, Z. Chen, E.S. Silveira, L. Zhang, M. Wei, C. Liu, C. de Sena-Tomas, K. Targoff and W. Min
Nat Commun. 2018; 9: 2995. Published online 2018 Aug 6. doi: 10.1038/s41467-018-05401-3
Multicolor Activatable Raman Probes for Simultaneous Detection of Plural Enzyme Activities
H. Fujioka, J. Shou, R. Kojima, Y. Urano, Y. Ozeki and M. Kamiya
J. Am. Chem. Soc. 2020, 142, 49, 20701–20707, Publication Date: November 23, 2020.
Metabolic activity induces membrane phase separation in endoplasmic reticulum.
Shen Y, Zhao Z, Zhang L, Shi L, Shahriar S, Chan RB, Di Paolo G and Min W.
PNAS 2017, 114(51):13394-13399. DOI: 10.1073/pnas.1712555114
Supermultiplexed optical imaging and barcoding with engineered polyynes.
Hu F, Zeng C, Long R, Miao Y, Wei L, Xu Q, Min W.
Nat Methods. 2018 Mar;15(3):194-200. doi: 10.1038/nmeth.4578. Epub 2018 Jan 15.
Applications of vibrational tags in biological imaging by Raman microscopy.
Zhao Z, Shen Y, Hu F, Min W.
Analyst. 2017 Oct 23;142(21):4018-4029. doi: 10.1039/c7an01001j.
Super-multiplex vibrational imaging
Lu Wei, Zhixing Chen, Lixue Shi, Rong Long, Andrew V. Anzalone, Luyuan Zhang, Fanghao Hu, Rafael Yuste, Virginia W. Cornish and Wei Min
Nature. 2017 Apr 27; 544(7651): 465–470. Published online 2017 Apr 19. doi: 10.1038/nature22051
Chemically-activatable alkyne-tagged probe for imaging microdomains in lipid bilayer membranes
Satoshi Yamaguchi, Taku Matsushita, Shin Izuta, Sumika Katada, Manami Ura, Taro Ikeda, Gosuke Hayashi, Yuta Suzuki, Koya Kobayashi, Kyoya Tokunaga, Yasuyuki Ozeki and Akimitsu Okamoto
Sci Rep. 2017; 7: 41007. Published online 2017 Jan 24. doi: 10.1038/srep41007
Early CRS Publications
Video-rate molecular imaging in vivo with stimulated Raman scattering.
B.G. Saar, C.W. Freudiger, J. Reichman, C.M. Stanley, G.R. Holtom, X.S. Xie
Science. 2010 Dec 3;330(6009):1368-70. doi: 10.1126/science.1197236.
Coherent Raman Scattering Fiber Endoscopy.
B.G. Saar, R.S. Johnston, C.W. Freudiger, X.S. Xie, E.J. Seibel
Opt Lett 36 (2011) 2396–2398.
Picosecond spectral coherent anti-Stokes Raman scattering (CARS) imaging with principal component analysis of meibomian glands.
C.Y. Lin, J.L. Suhalim, C. Nien, M.D. Miljkovic, M. Diem, J. Jester and E.O. Potma
J Biomed Opt 16 (2011) 021104.
Fast and long term lipid droplet tracking with CARS microscopy.
C. Jüngst, M. Winterhalder, A. Zumbusch
J Biophot 4 (2011) 435–441.
Quantitative detection of chemical compounds in human hair with coherent anti-Stokes Raman scattering.
M. Zimmerley, C-Y. Lin, D.C. Oertel, J.M. Marsh, J.L. Ward and E.O. Potma
J Biomed Opt 14 (2009) 044019.
Coherent anti-Stokes Raman Scattering Microscopy.
M. Müller, A. Zumbusch
ChemPhysChem 8:15 (2007) 2156–2170.
Nonperturbative chemical imaging of organelle transport in living cells with coherent anti-stokes Raman scattering microscopy.
X. Nan, E.O. Potma and X.S. Xie
Biophys J 91 (2006) 728–735.
Coherent anti-stokes Raman scattering microscopy.
X.S. Xie, J.X. Cheng and E. Potma
Handbook of biological confocal microscopy, 595-606 (2006).
Chemical imaging of tissue in vivo with video-rate coherent anti-stokes Raman scattering microscopy.
C.L. Evans, E. Potma, M. Puoris’Haag, D. Côté, C.P. Lin and X.S. Xie
Harvard University, Cambridge, MA, September 22, 2005.
CARS for Biology and Medicine.
E.O. Potma and X.S. Xie
Optics and Photonics News 15 (2004) 40–45.
Real-time visualization of intracellular hydrodynamics in single living cells.
E.O. Potma, W.P. de Boeij, P.J. van Haastert and D.A. Wiersma.
Proc Natl Acad Sci USA 98 (2001) 1577–1582.
Vibrational Microscopy Using Coherent Anti-Stokes Raman Scattering.
A. Zumbusch, G.R. Holtom, X.S. Xie
Phys Rev Lett 82 (1999) 4142.
Coherent anti–Stokes Raman spectroscopy.
R.F. Begley, A.B. Harvey and R.L. Byer
Applied Physics Letters 25:7 (1974), Microwave Laboratory, Stanford University, Stanford, California 94305.
Study of Optical Effects Due to an Induced Polarization Third Order in the Electric Field Strength.
P.D. Maker, R.W. Terhune
Physical Review 137:3A (1965) 801–818.