Virology
In early 2020, the field of virology was suddenly thrust into the public consciousness after the outbreak of SARS-CoV-2, a virus that causes a novel respiratory disease in humans. This disease, called COVID-19, caused a global pandemic. As a result, there have been monumental efforts to treat the disease and limit its spread.
Understanding the behavior of these infectious agents is fundamental to our ability to treat viral diseases and prevent further outbreaks. Virology seeks to understand various aspects of viruses, including their structure, evolution, entry into host cells, reproduction, the diseases they cause, how organisms respond to infection, and their spread between individuals.
One of the crucial aspects of designing and implementing effective treatments and preventative measures is to be able to visualize viral components and their effects on host cells. For this visualization, immunofluorescence microscopy is a valuable methodology.
Basics of immunofluorescence
Immunofluorescence [1,2] is a method in which the binding specificity of antibodies to their antigens are used to visualize the structure and dynamics of biological systems. Antibodies are raised against specific proteins and either directly tagged with a fluorescent marker or used in conjunction with fluorescently tagged secondary antibodies to identify and study the presence of the protein of interest.
This broadly used and versatile technique allows scientists to visualize proteins, cell components, and cell processes in a variety of contexts. For example, it can be used to study the structure of organelles, the distribution of proteins in a tissue, and stain for the presence of specific biomarkers in a sample. How can immunofluorescence be employed in the performance of virology research?
Immunofluorescence in virology research
Viruses are nanoscopic agents with most less than 0.4 µm in size; an electron microscope is required to visualize their nanostructure. However, standard and super-resolution microscopy techniques are robust and accessible tools that can be put to use for understanding the larger scale structure and function of viruses.
Immunofluorescence is compatible with a variety of imaging approaches, including standard widefield imaging, confocal microscopy, lifetime imaging, and super-resolution microscopy [3-4]. The choice of which to use largely depends on the specific application.
Diagnosis
Standard immunofluorescence staining with widefield microscopy has long been used to diagnose viral infections such as influenza from nasopharyngeal swabs from patients. An antibody against a virus-specific protein is used to stain the tissue of interest, and a clinician can quickly determine whether the virus is present in the sample. This procedure lacks the sensitivity of more advanced molecular methods; however, it is quick, simple, and clinically valuable [5,6].
Mechanism of infection
One crucial feature of viruses is that they must enter host cells to reproduce and cause disease. Once they attach to the host cell, they release their genetic material inside. The genetic material is recognized and expressed by the cell. The encoded components are then present in multiple copies, which are then able to assemble into new viral particles. These new particles are released by the cell and can go on to infect new hosts. Thus, the virus hijacks the host cell machinery to promote its pathogenic effects.
Being able to understand this complex and highly variable process is critical for understanding the infection mechanisms of viruses, especially more recent viruses like SARS-Cov-2. For example, immunofluorescence assays have been utilized to study the binding dynamics of SARS-Cov-2 to cell surface receptors [7] and to study its intracellular dynamics, including how the viral particles assemble inside the cell [8].
Effects on cell function
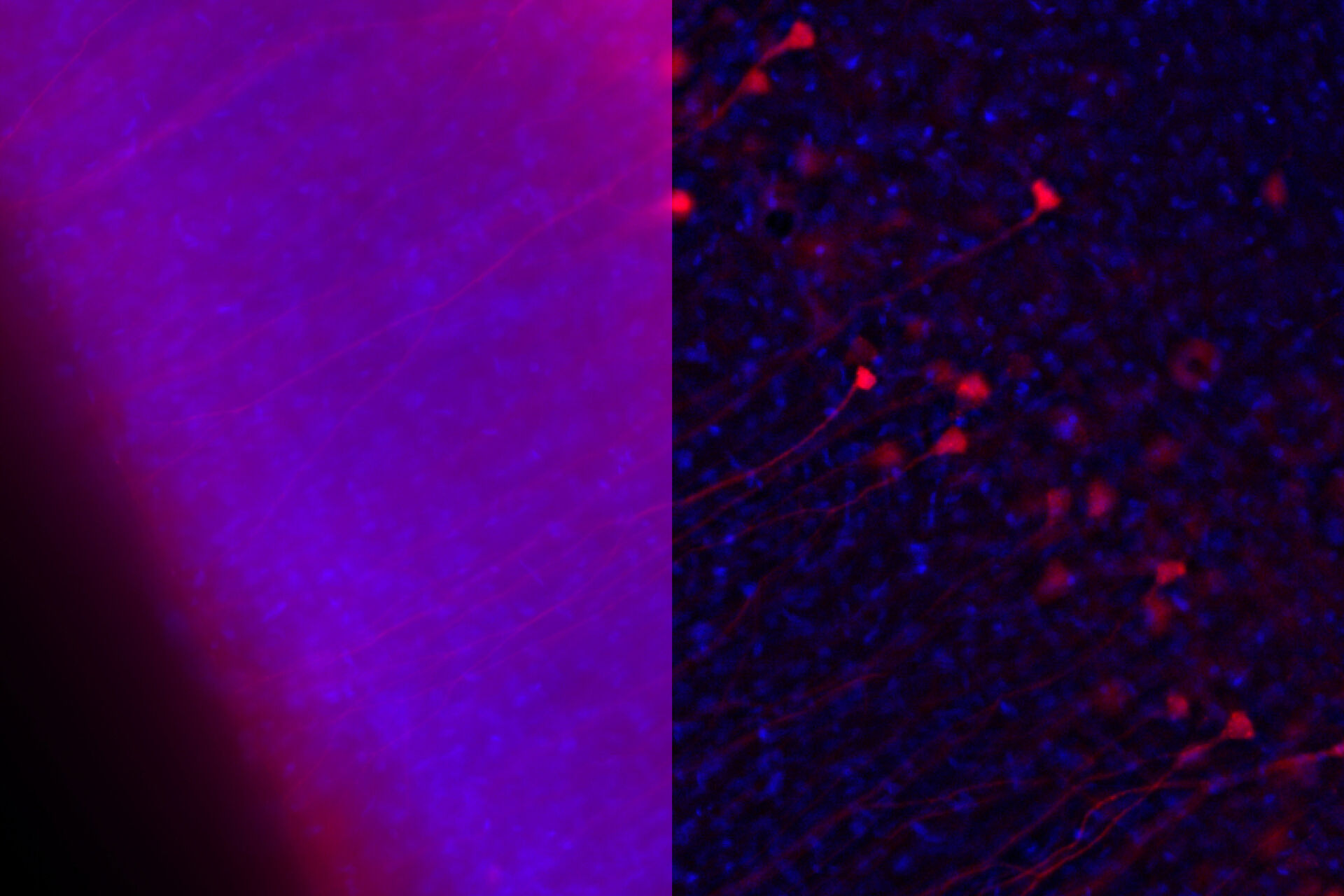
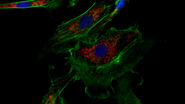
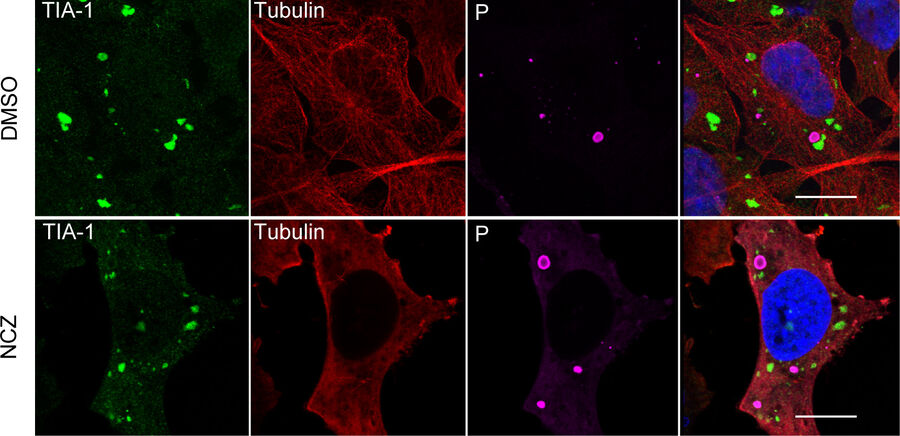
Along with detecting the presence and revealing the dynamics of viral invasion, it is also crucial that we understand the pathogenesis of the infection. Immunofluorescence is a valuable asset for these investigations, as being able to visualize the cell response is crucial for understanding the viral disease. Such avenues of study could include the dynamics of the cellular stress response to viral infection [9], and how viruses impact cell and tissue repair [10]. For example, one study showed that rabies virus infection of cells can promote the formation of cytoplasmic stress granules, membrane-less dynamic structures consisting of mRNA and protein aggregates that form rapidly in response to cellular stresses [9]. The researchers recorded images of immunofluorescently stained cells using a Leica confocal microscope. The other study revealed that the repair of epithelial stem/progenitor cells infected with an influenza virus (IV) critically depended on fibroblast growth factor receptor 2b (Fgfr2b) signaling [10]. Some images of immunofluorescently stained cells were taken with a Leica upright microscope (refer to figure 1).
Treatments
Finally, in order to design and deliver safe and effective therapies and preventative vaccines, we must observe how the host cells – and their interactions with the virus - are affected by treatments. Immunofluorescence can be employed to test the localization and behavior of antiviral agents inside cells [11], the binding characteristics of antibody cocktails [12], and the effect of treatments on the presence and localization of viral particles inside the cell.
Conclusions
While molecular studies are an essential tool in the field of virology, microscopy provides indispensable insights for research and clinical applications helping to complete the picture showing the relation between function and structure. Immunofluorescence assays are usually quick and straightforward procedures that enable researchers to visualize the effects and dynamics of viral infections. In the current global climate after the occurrence of the COVID-19 pandemic, such methods represent a crucial tool in the prevention and treatment of these pathogens.