Workflow for checking cell confluency
Checking cell confluency is a routine part of cell culture workflows commonly used for diverse applications in life science research and in biopharmaceutical-industry laboratories [1,2]. Monitoring cell growth is essential whether researchers are propagating their culture or preparing their cells for further transfection, differentiation, wound healing, clone selection, colony picking, or 3D culture of organoids [3,4].
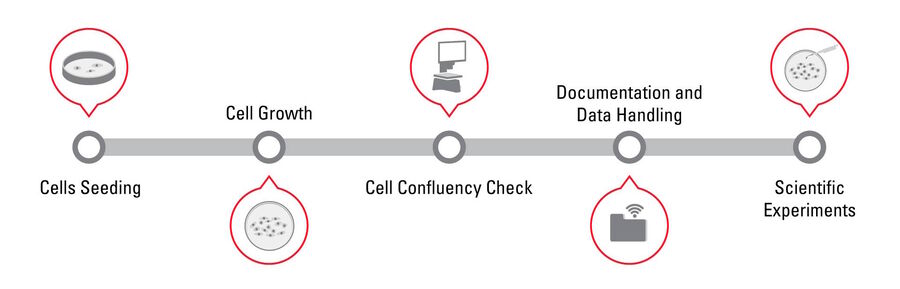
Confluency Check Workflow Steps: 1. Cell Seeding, 2. Cell Growth, 3. Cell Confluency Check, 4. Data handling, and 5. Scientific experiments [5,6].

Step 1: Cell seeding
Seed the cells on the preferred cell culture vessel and place them in the incubator. Note that cell seeding density can determine the proliferation of the culture.

Step 2: Cell growth
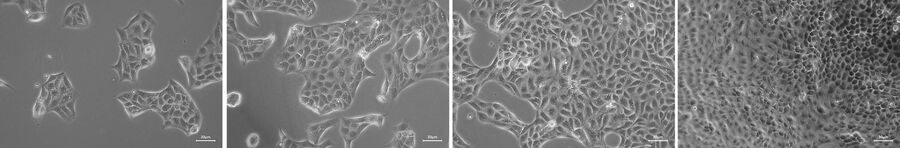
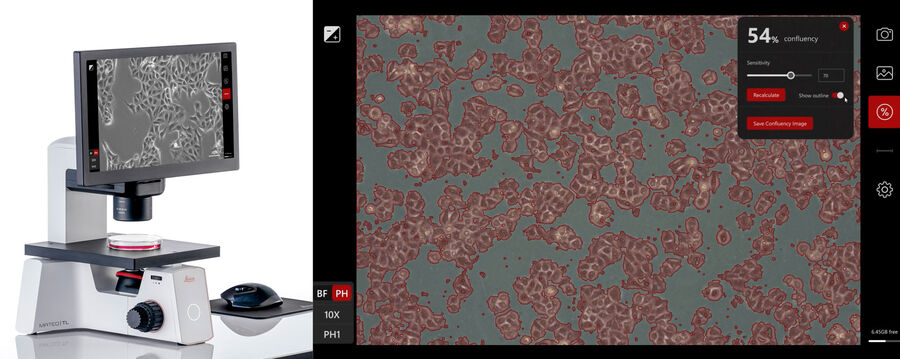
Let the cells settle down, attach to the plate, and start growing. Observation of cell morphology and growth can be done using Mateo TL in phase-contrast mode (Figure 1) and facilitated by the assisted contrast setup. Document the status of the cell culture by capturing images.

Step 3: Checking cell confluency
In cell culture, confluency is the percentage of the surface area covered by a layer of cells in a culture vessel [2].
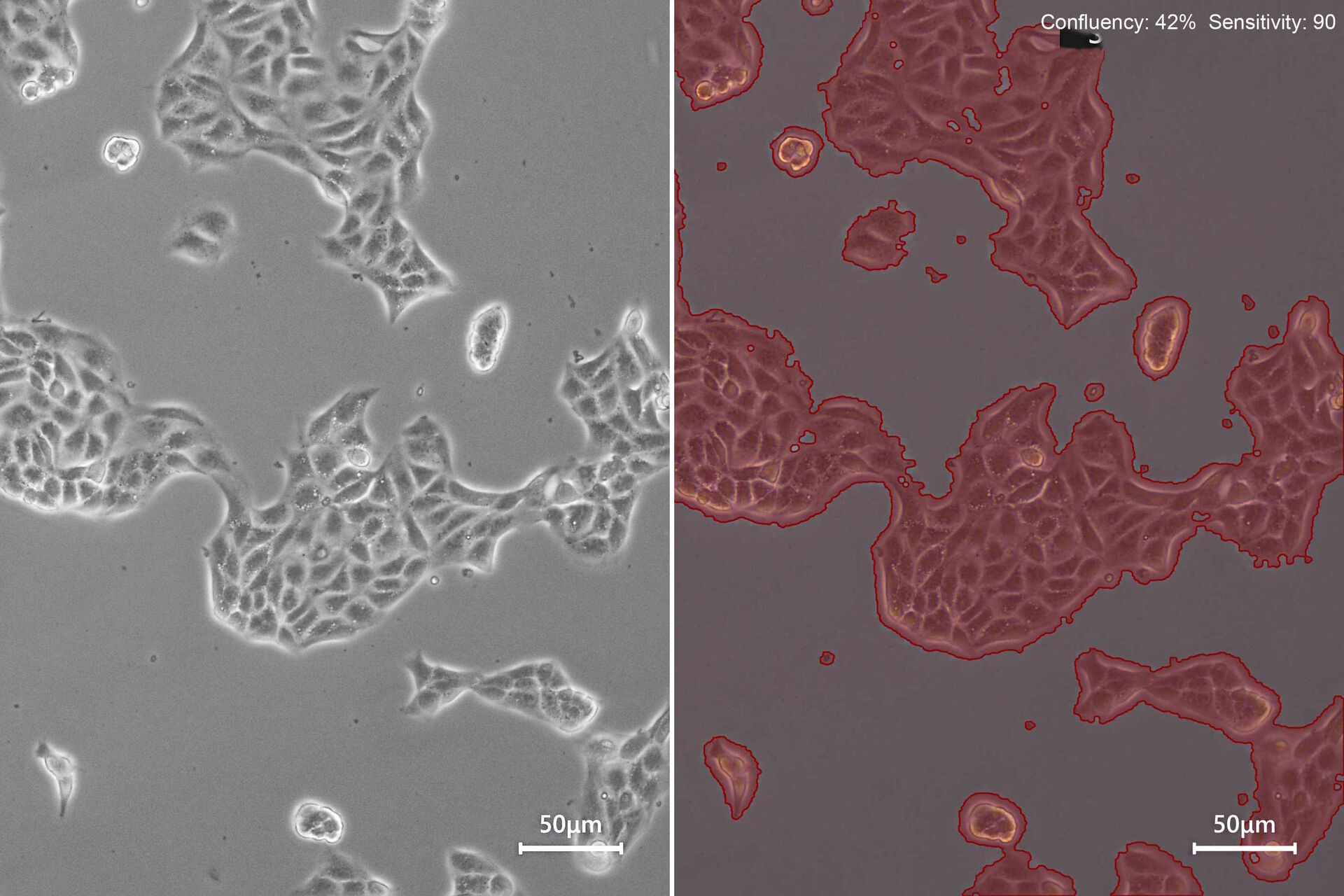
Check how confluent the cell culture is with the Mateo TL confluency module. Adjust the sensitivity of border detection and apply the confluency measurement.
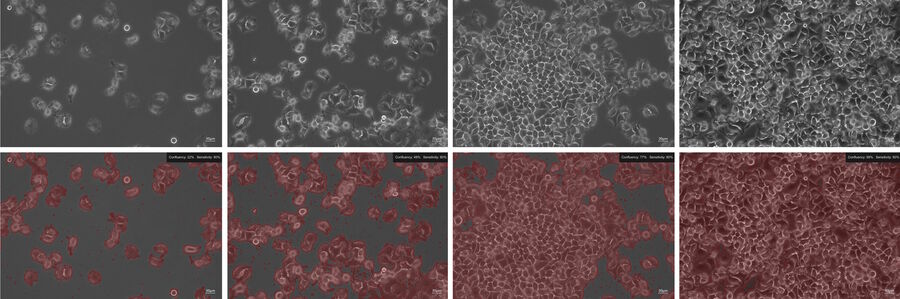
The Mateo TL confluency algorithm automatically calculates the percentage area covered by cells in the image. Because cells can be unevenly distributed across growth in multiple areas, it is recommended that the confluency be calculated at a low magnification and different areas of the culture vessel.
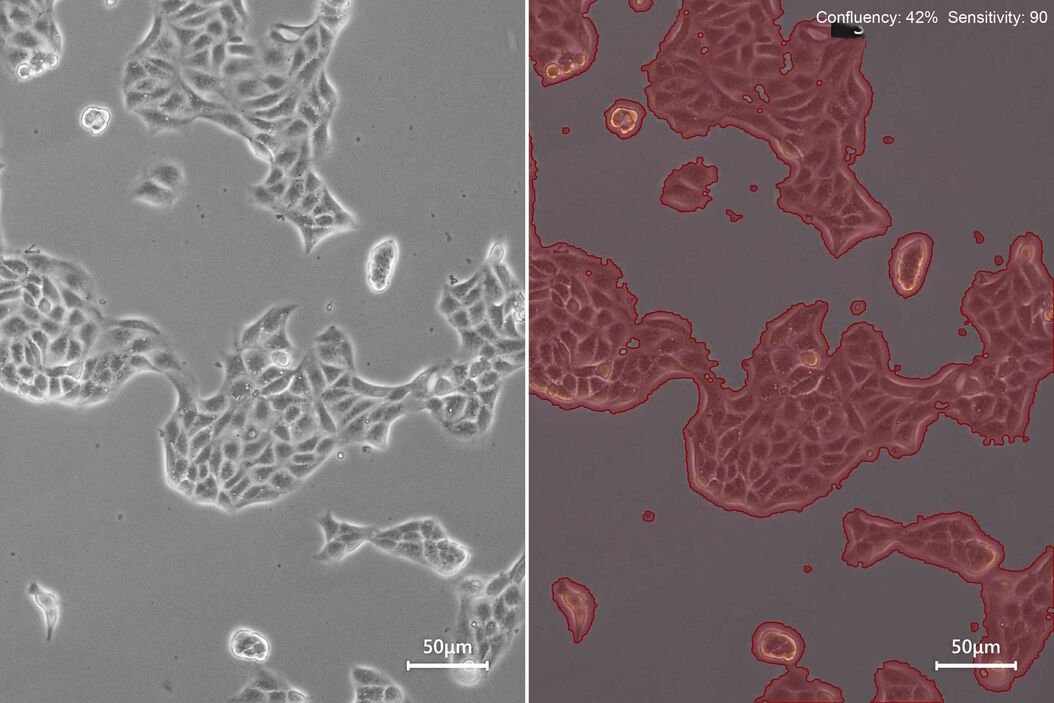
For reporting and cell-culture tracking, it is recommended to save two images: the image of the current cell- growth status using phase contrast (Figure 3, top) and the image generated by the confluency calculation (Figure 3, bottom).

Step 4: Documentation and data handling
After the cell culture status has been documented using images, and the cell confluency has been measured, images can be quickly and conveniently exported using enhanced smart functionalities.
Images can be wirelessly transferred directly to your smartphone or tablet with the quick share feature or exported to USB.
If image acquisition needs to be repeated, then the imaging conditions, i.e., light and camera settings, of a previous reference image in the gallery can be easily reproduced using the repeat function.
Step 5: Scientific experiments
After documenting the current culture status and measuring the cell confluency with the Mateo TL confluency module, it is time to decide how to proceed further.
Routine cell-culture maintenance, such as changing the media, keeping cells in culture, expanding or sub-culturing cells, stocking, or stopping the culture are just few examples.
Or, if the culture has reached the desired confluency status, then the experiment can be started or proceed to the next step. Having a specific cell culture confluency as a starting point is essential for assays, such as gene transfection, clone selection, cell line development, colony picking, and reprogramming and differentiation.
For colony picking or other protocols requiring simultaneous sample handling and microscopic observation, it is recommended to clean the Mateo TL microscope and operate it inside the laminar flow cabinet. Its digital display eliminates the use of oculars, minimizes fatigue, and enables its use inside the flow hood, minimizing contamination risks (Figure 4).
Mateo TL Digital Transmitted Light Microscope
Routine checks of cell culture are now easier and consistent.
Mateo TL enables an easy cell culture check and consistent confluency evaluation:
- Ready-to-use microscope: Go from setup to first image in under one minute.
- Facilitate routine confluency checks and standardize the search for the starting time points for experiments with the Confluency Module.
- Achieve consistent confluency measurements between different users and experiments with one common criterion for measurement.
- Minimize human bias by taking the guesswork out of confluency evaluation.
Routine cell culture? Check!
Disclaimer: This article describes a generic workflow for a quick cell confluency check. Please feel free to use the recommended information to help improve your scientific experiments. Also, please note that the information in the article does not replace the cell-culture guidelines or SOPs used in your laboratory, nor your technical expertise or knowledge. Leica Microsystems cannot be held responsible for unexpected or undesired outcomes resulting from experiments which are performed in accordance with the information reported here.