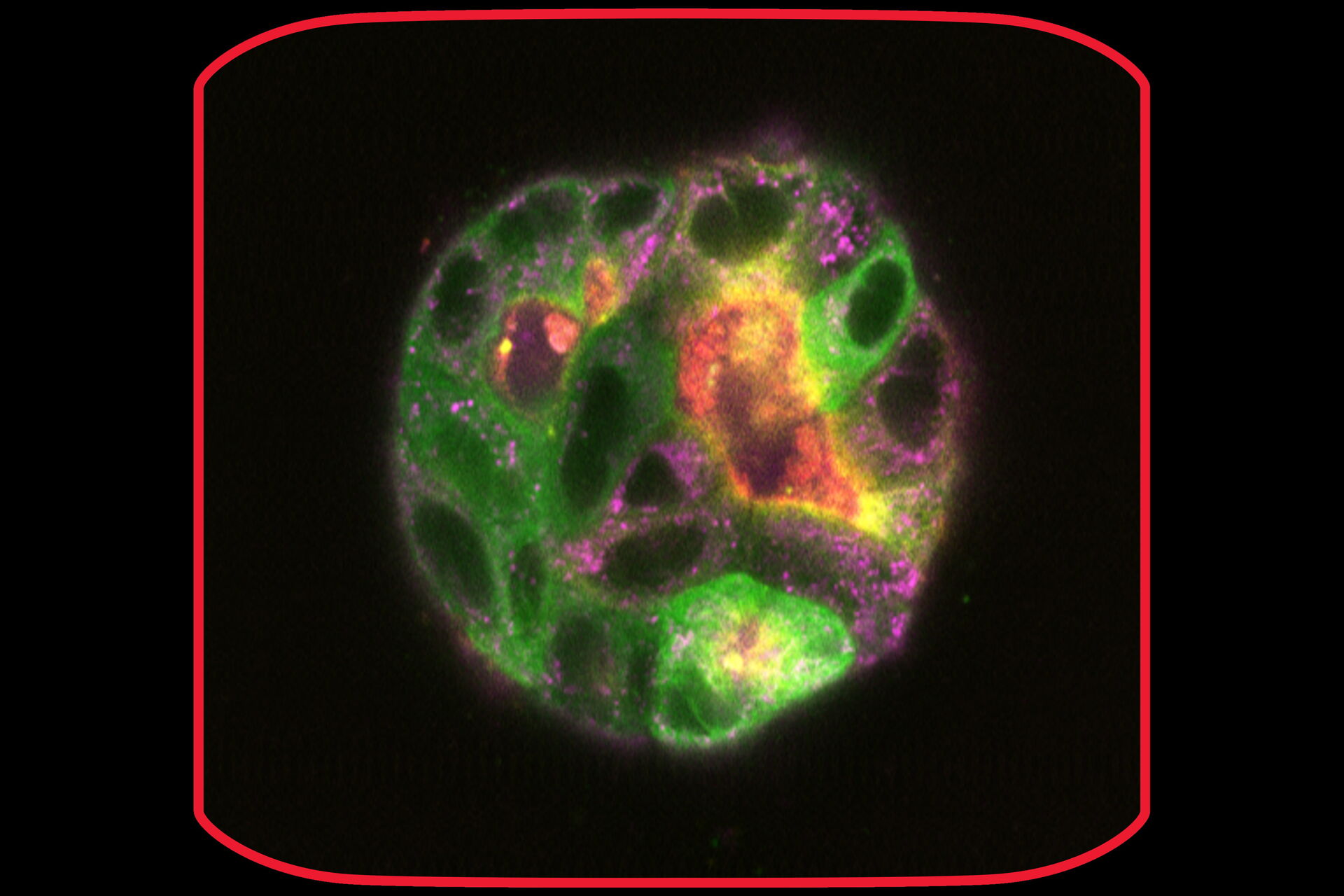
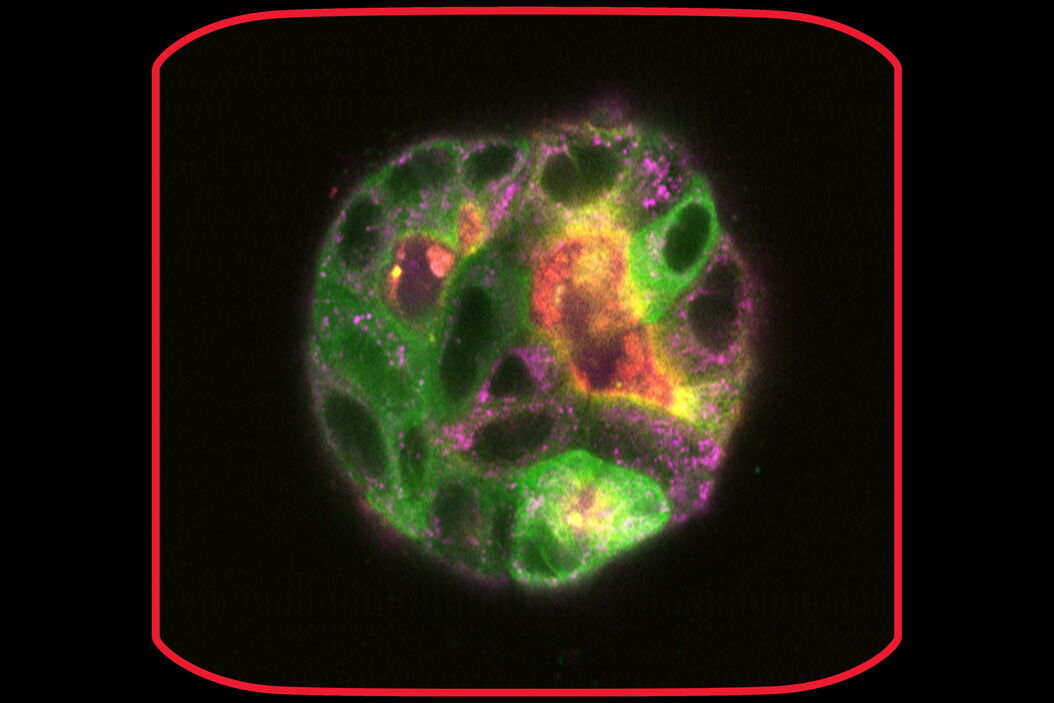
In the following paragraphs, there are a few examples of live-cell imaging with Mica. See how you can get reliable results ranging from fast, short-term experiments up to long-term ones taking place for weeks. For instance, Mica allows imaging of typical specimens such as monolayer cell cultures to more complex live tissues, organoids, or model organisms. Moreover, the integrated incubation system delivers physiological-like conditions concerning temperature and pH, enabling live-cell imaging for weeks.
Example: Short-term experiment with fast events
U2OS cells were labelled with two different fluorescence stains marking the same structure, WGA-Alexa488 and WGA-Alexa555 which both bind to cellular membranes. By acquiring both channels in Mica sequential mode, this experimental setup enables to introduce an artificial time shift between the acquisition of WGA-Alexa488 (green) and WGA-Alexa555 (red). With it, the same membrane structures, which are marked with the two different Alexa dyes are imaged at two minimally shifted timepoints. In the comparison of both methods, you can see that while in the simultaneous recording all vesicles are yellow – a mixture of green and red – in the sequential recording some fast moving vesicles appear to be two different structures – one green and one red vesicle.