Fit for the environment of an electron microscope
The chamber of an electron microscope is evacuated to a very low pressure. The structure of a living cell placed into this environment cannot be preserved due to the extremely quick evaporation of the water which makes most part of the cell.
There are a number of preparation possibilities for biological samples. The material could be preserved (fixed) so the subsequent dehydration produces minimal damage to the in vivo structure, an environmental SEM could be used or the water could be frozen. High pressure freezing is the only way to observe hydrated structures in their natural state. The ice built by high pressure freezing is not hexagonal ice, which shows an increase of volume from water to ice but amorphous ice, where the volume stays constant. Therefore structures which are sensitive to osmotic and temperature change are preserved (see the article "Brief introduction to High Pressure Freezing").
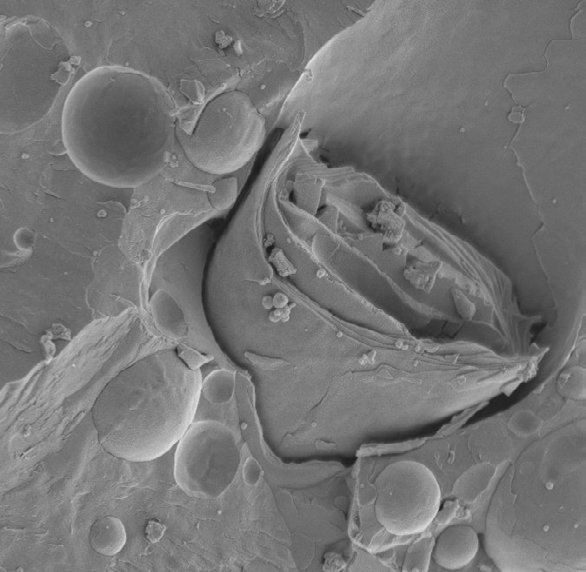
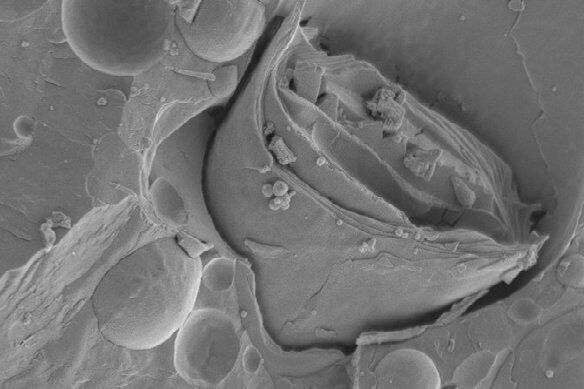
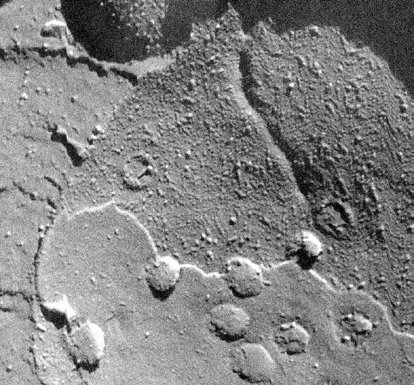
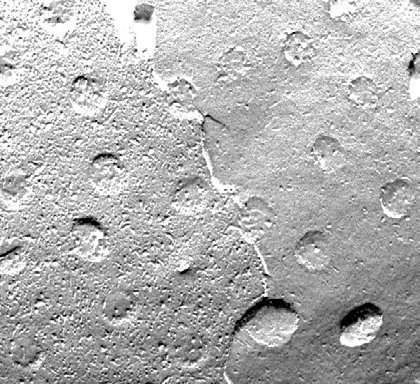
To observe structures such as cell organelles, membranes, emulsions or surface interfaces of liquids, freeze fracture is the only way to do that. The frozen sample is broken by force of a knife (or similar) or a released spring load and breaks along the lines of least resistance.
Fig. 2: Freeze fracture (Source: http://en.wikibooks.org/wiki/Structural_Biochemistry/Lipids/Membrane_Fluidity)
Sublimation and condensation of water – Freeze etching and contamination
To reveal details of the fractured surface, ice has to be removed. This needs to be done by sublimating the ice to preserve the structure of the specimen. Ice is directly transformed into water vapour without going through the liquid state which would lead to a change in volume and structural damage.
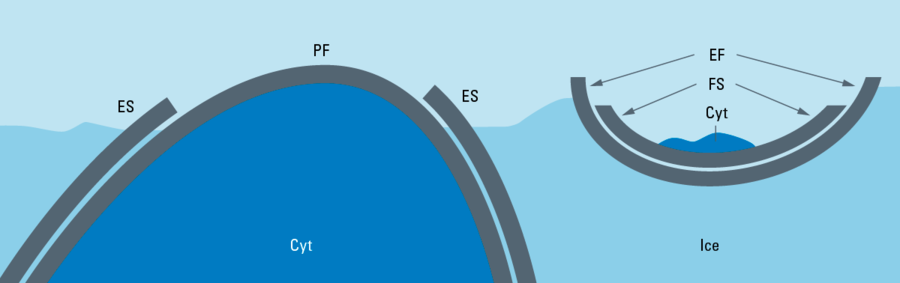
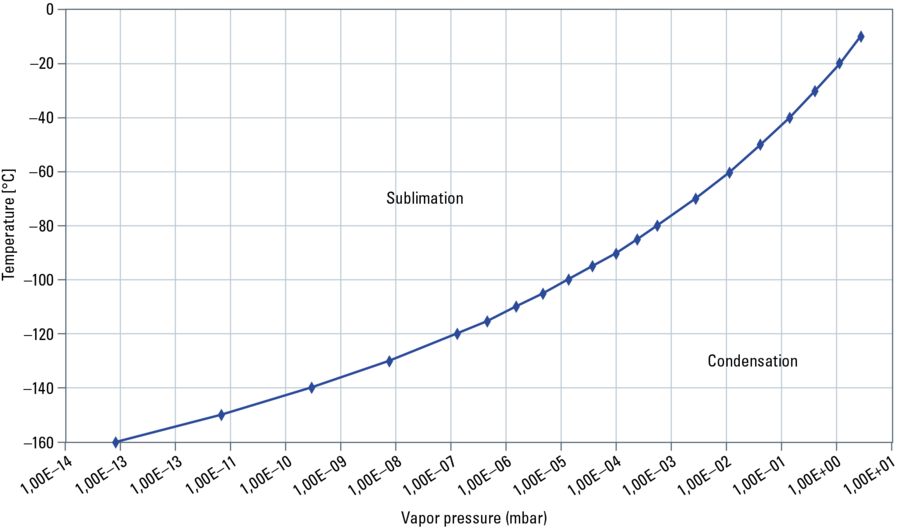
The sublimation/condensation process of water depends on the saturation pressure at a particular temperature and the effective partial water pressure of water or ice in the chamber. Note: a good vacuum reduces the partial water pressure.
For example: Ice or frozen specimens with a temperature of –120 °C have a saturation pressure of about 10–7 mbar. If this pressure is established in the chamber, condensation and evaporation are in equilibrium. The amount of evaporated molecules is equal to the amount of condensed molecules. At a higher pressure the condensation rate is higher than the sublimation rate – ice crystals grow on the specimen’s surface. This has to be avoided by all means. A colder (than the specimen) plate above the specimen reduces the local pressure and works as a condensation trap. Water molecules driven up from the specimen preferentially attach to the colder surface. At a lower pressure than the saturation pressure more molecules sublimate than condensate and freeze etching takes place.
Performing freeze etching until the sample is completely ice free, is called freeze drying. This process only works for small samples to be performed in a reasonable time. It is done in several steps by heating up from around –120 °C to –60 °C maintaining the temperature of each step for a certain time. This can take up to days.
At specimen temperatures below –120 °C the etching rate is very low, etching times increase to impractical durations. If the pressure of the vacuum chamber is fixed, it is possible to increase the etching rate by raising the specimen temperature. Careful with temperatures higher than –90 °C for biological samples. Etching rates increase tremendously. Additionally hexagonal ice forms from the vitrified ice and causes dehydration artefacts.
The theoretical sublimation rates of pure water are reduced because:
- Water from the depth of a specimen sublimates slower than water from the surface.
- Solvents of salt and macromolecules reduce the sublimation speed.
- The bound water which is a considerable part of biological samples have a lower sublimation rate.
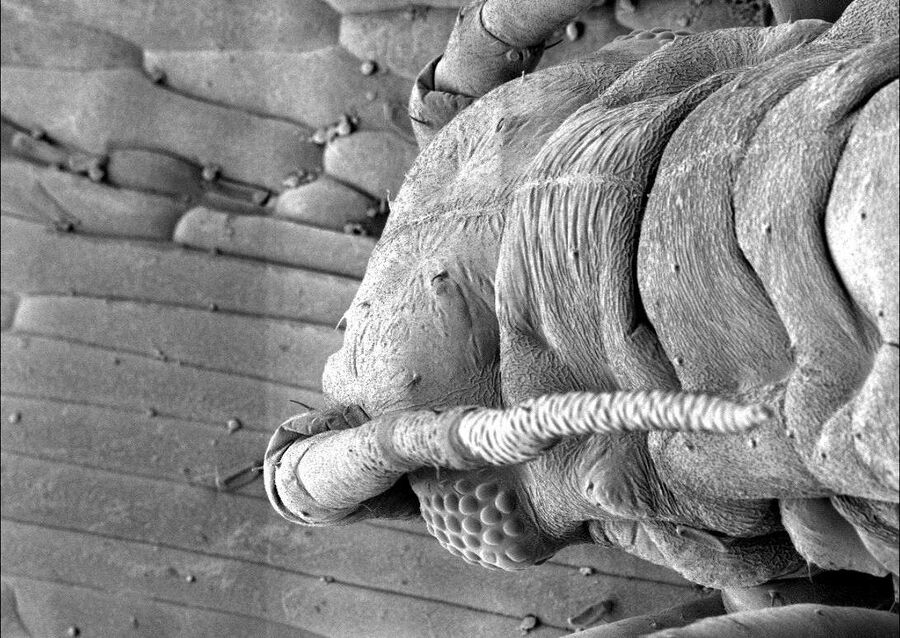
Freeze fracture to generate images
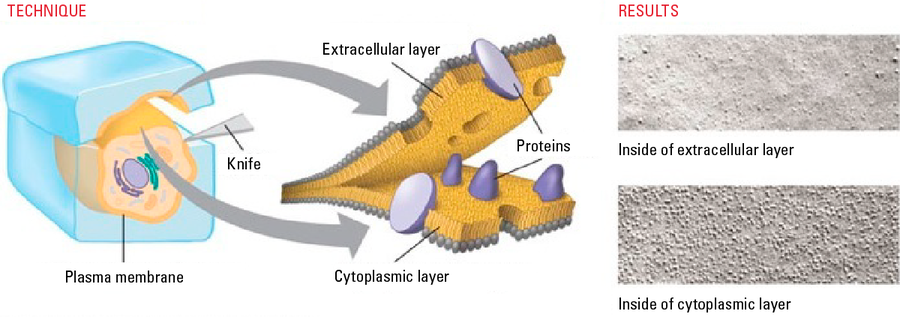
Freeze-fracture and freeze etching techniques require ultrathin heavy metal and carbon films deposited under vacuum on the fractured surface.
Freeze fractured samples are coated under an angle with metal followed by a carbon backing up film (Leica EM ACE600 freeze fracture with Leica EM VCT500) to produce either a replica to be imaged in a TEM or a block face for the SEM.
For both methods the fractured surface is coated after a certain amount of etching time the same way. First a thin (2–7 nm) heavy metal coating under an angle to produce topographic contrast (shadowing). Second a thick carbon layer (15–20 nm) coated under 90° to stabilize the ultra-thin metal film. At this point the etching process is stopped. To image very small structures the heavy metal is applied in a very low angle (2–8°) and the sample is rotated during coating. This adds contrast to filamentous and small structures. Such technique is called low angle rotary shadowing.
E-beam evaporation should be used for the heavy metal film. This is the coating technique giving a very fine and directional deposition. The supporting layer of carbon stabilizes the structures which are uncovered by metal. Those structures would change their contour during the increase of temperature, the sample would not be completely conductive and a replica would not stick together.