The problem: Data integrity at risk
Laser microdissection (LMD) isolates specific regions of interest or cell populations from tissue sections using a focused UV laser. In gravity-based systems like the Leica LMD7, dissected fragments fall into collection wells below. Precise molecular analysis depends on samples reaching their intended destinations.
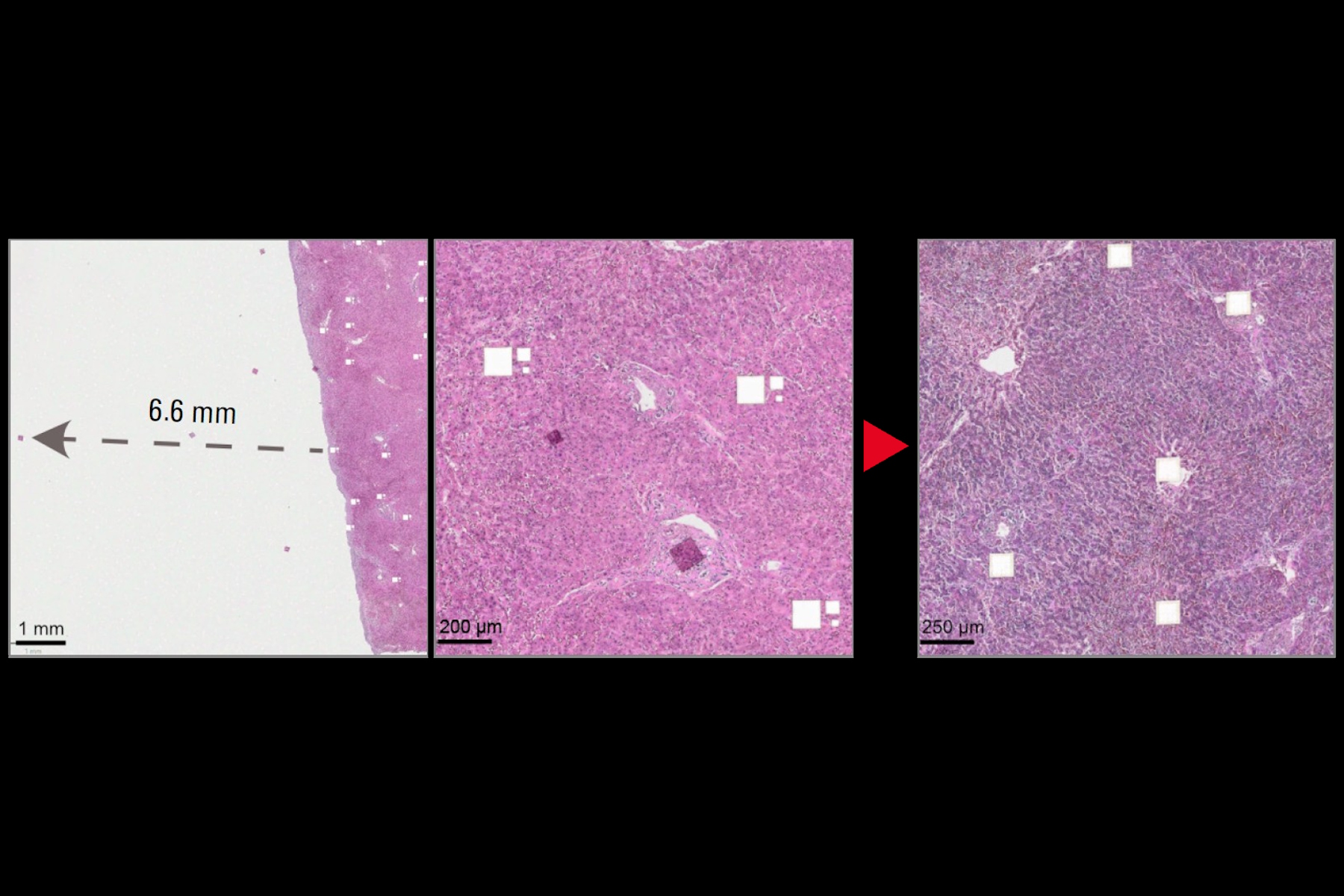
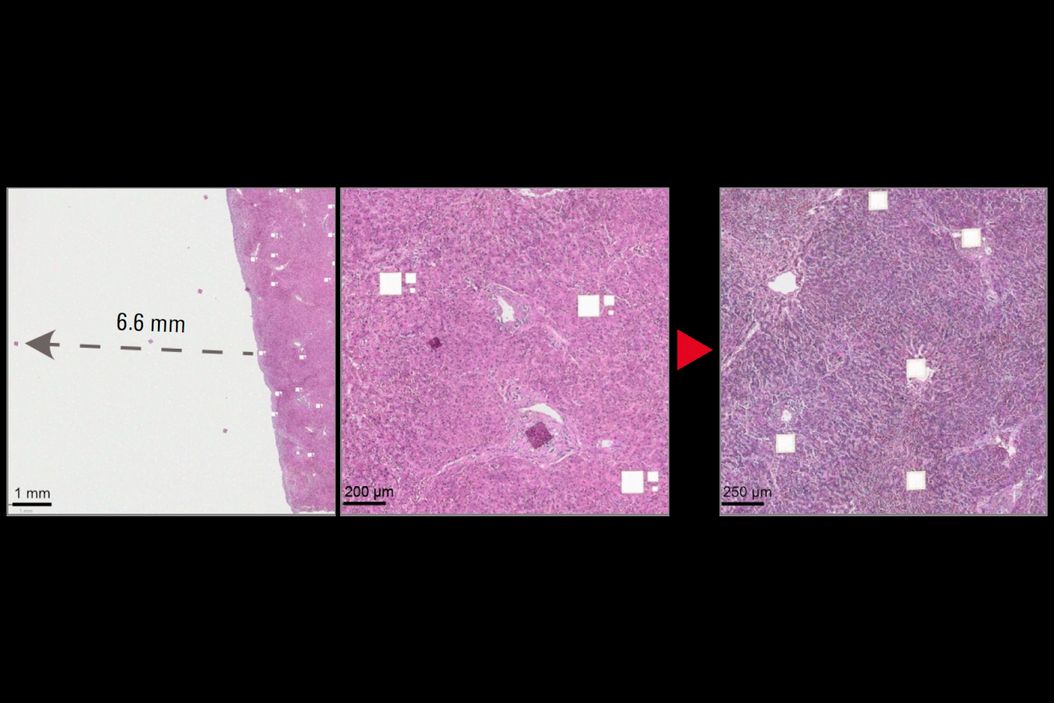
Static charge accumulates on slides, membranes, collection devices, and chamber walls. This causes two distinct failures. First, fragments adhere to slides or frames and never reach collection wells—samples are lost. Second, fragments fly laterally into adjacent wells—leading to cross-contamination and generation of false discoveries. We measured fragments traveling up to 6.6 mm off-target, sufficient to reach adjacent wells in a 384-well plate. Cross-contamination is particularly dangerous: unlike lost samples, contaminated wells produce false results without warning.
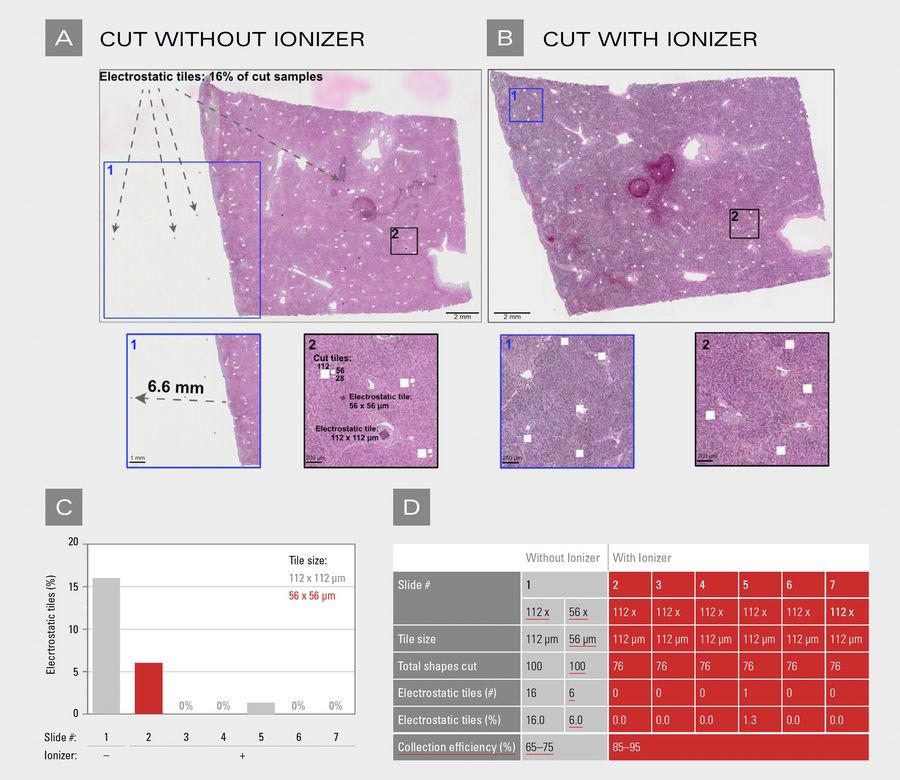
Without intervention, 16% of cuts resulted in electrostatic problems (16/100 cuts in our validation): some samples lost, others displaced into wrong wells.
Current solutions fail. Manual anti-static guns provide momentary relief before charge rebuilds. Humidity control conflicts with protocols requiring dry conditions and lacks precision. Researchers must choose between compromised efficiency and visual verification of every cut.
The solution: Integrated ionization
We positioned a bar-type ionizer with integrated ground-plate technology inside the LMD environmental chamber. The critical design choice: operating with the chamber closed. Ions equilibrate throughout the enclosed volume, neutralizing charge on all surfaces including plastic chamber walls. Result: a single electrostatic event in 450+ cuts (0.2%), effectively eliminating both sample loss from adhesion and cross-contamination from lateral displacement.
Installation
Mount the ionizer bar on the environmental chamber at 45° from horizontal, 100–200 mm from the sample stage. Use cable ties or non-conductive fasteners. Ensure a clear line of sight between the ionizer bar and the sample stage to insure efficient ion delivery.
Set frequency to 22–68 Hz for this distance range. Allow 30–60 minutes pre-equilibration with the chamber closed before cutting. This period fills the enclosed volume with ions and neutralizes all surfaces. No compressed air required.
Results
| Metric | Without Ionizer | With Ionizer |
|---|---|---|
| Cuts performed | 100 | 450+ |
| Electrostatic displacement events | 16 events (16%) | 1 event (0.2%) |
| Maximum sample displacement | 6.6 mm | None |
| Collection success rate | 65–75% | 85–95% |
| Cross-contamination risk | Present | Effectively eliminated |
Table 1: Performance comparison with and without ionizer integration
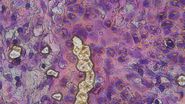
The ionizer eliminated erratic fragment behavior. Samples released cleanly from membranes and fell directly into target wells (Figure 1). The single event occurred early in testing; subsequent refinement of equilibration time prevented recurrence. Importantly, this improvement was consistent across all three tile sizes tested (112 × 112 µm, 56 × 56 µm, and 28 × 28 µm), indicating that ionization is effective independent of fragment size (Figure 1A, B).
Collection success (85–95%) remains below 100% due to incomplete laser cuts, membrane adhesion, or well-edge effects. These losses are independent of electrostatic charge. The ionizer eliminates electrostatic losses—both samples stuck to charged surfaces and samples deflected into wrong wells.
A) H&E-stained liver slide 1 was cut in the LMD without an ionizing bar. Three different tile sizes were cut: 112 × 112 µm, 56 × 56 µm, or 28 × 28 µm. Insets highlight electrostatically displaced tiles across the slide. Scale bar: 2 mm. Inset 1 scale bar: 1 mm; inset 2 scale bar: 200 µm.
B) H&E-stained liver slide 2 was cut in the LMD with an ionizing bar. 304 tiles of 112 × 112 µm were cut. Insets show complete absence of electrostatically displaced tiles. Scale bar: 2 mm. Inset 1 scale bar: 250 µm; inset 2 scale bar: 200 µm.
C) and D) Quantification of electrostatically displaced tiles under different ionizing conditions.
Conclusion
A bar-type ionizer with integrated ground-plate technology, closed-loop ion current control, and pulse AC generation—operated within a closed environmental chamber—reduces electrostatic events from 16% to 0.2%. This eliminates both failure modes: samples no longer stick to charged surfaces, and samples no longer fly into adjacent wells.
The approach requires minimal equipment modification and integrates into standard workflows after initial equilibration. A bar-type ionizer thus enables reliable collection of even the smallest tissue samples, ensuring each well contains only its intended material.