Case Description
A 56-year-old male developed sudden pain in the right eye and vision loss. The patient had no prior trauma or surgery and he had 20/20 vision in the left eye. He was referred for angle closure with vitreous in the anterior chamber.
The intraocular pressure was extremely elevated at IOP 56mmHg, treated down to 23mmHg on four medication classes. The patient was hoping for a quick recovery to return to work.
The objective of the surgery was to manage the dislocated cataract to achieve a long-term good result without having a lens dislocation in the future.
Pre-operative assessment
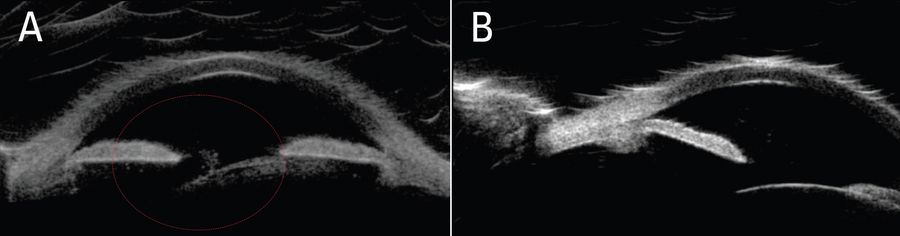
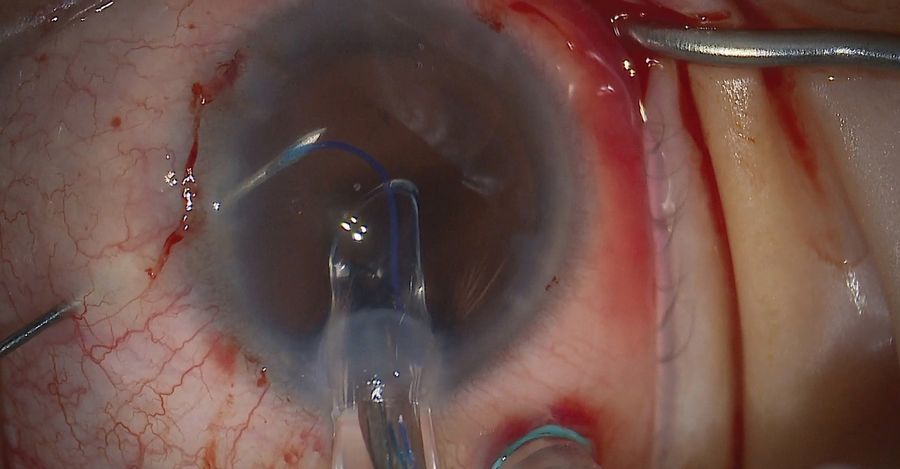
For the pre-operative assessment, intraoperative OCT scans revealed a tuft of vitreous in the anterior chamber and a severely tilted lens. There was no sulcus space on one side and the other side was hyper deep behind the iris, where the vitreous was coming around. The lens was very mobile, and the angle would open while the patient was supine (Figure 1).
Surgical approach
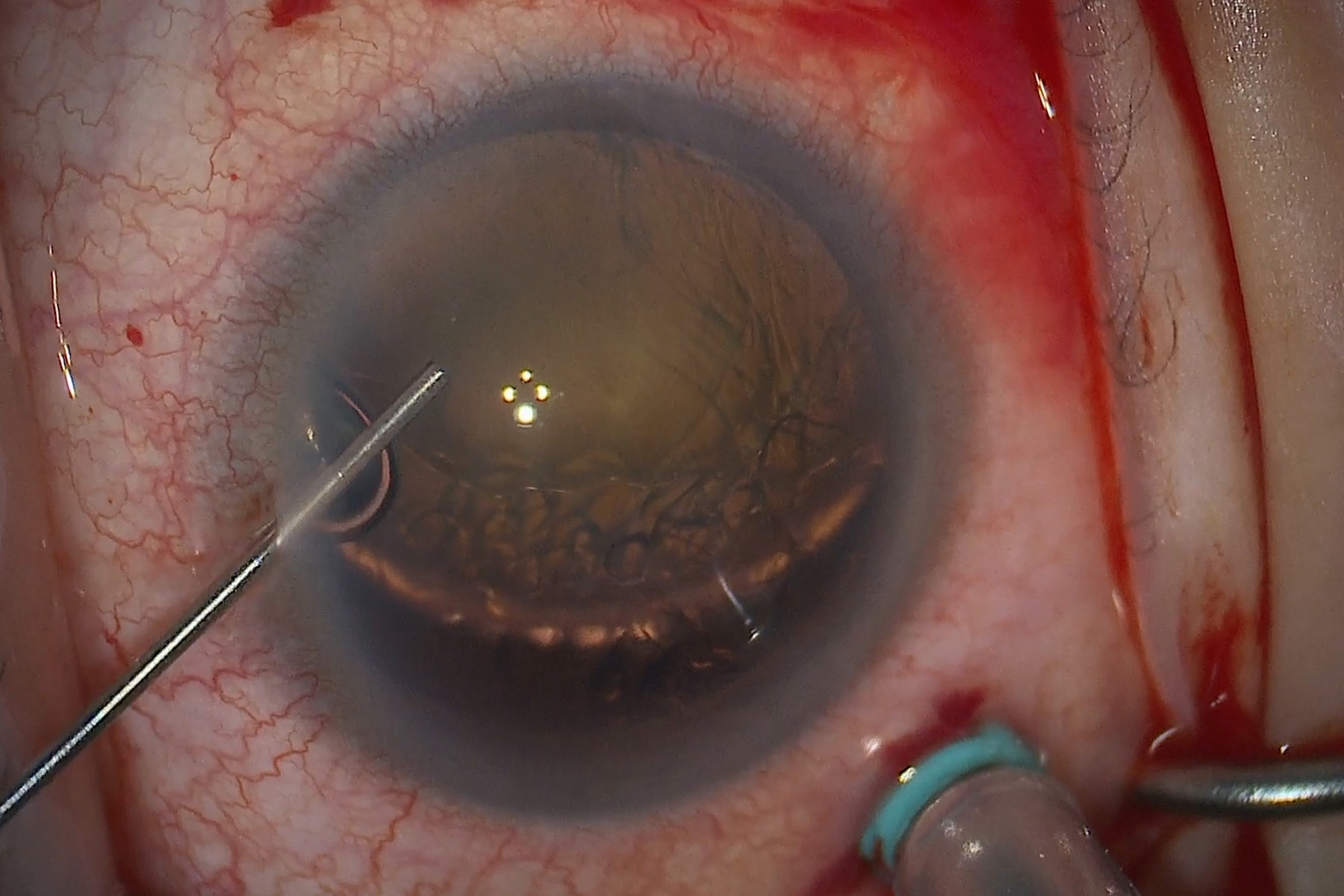
The lens was severely dislocated. A dispersive viscoelastic was applied to coat the anterior vitreous. A limited core vitrectomy was performed to draw the vitreous back and make sure it was not engaged in the phacoemulsification.
The zonules didn’t offer much resistance as the rhexis was made. Six iris hooks were used, which are a valuable tool in low flow states to keep the capsule on the lens elevated.
As the rhexis began, an iris hook was placed and the rhexis drawn around. When reaching a point with less countertraction, and to avoid a shift of the lens, a switch was made to capsule retractors for use during the cataract surgery.
Space was created with a cohesive viscoelastic to avoid trapping cortex. The capsule retractors were then placed in the corneal incision while using the viscoelastic injector to keep the lens elevated. The capsule retractors are much broader, which helps to stabilize the lens during phacoemulsification with less risk of radial tear.
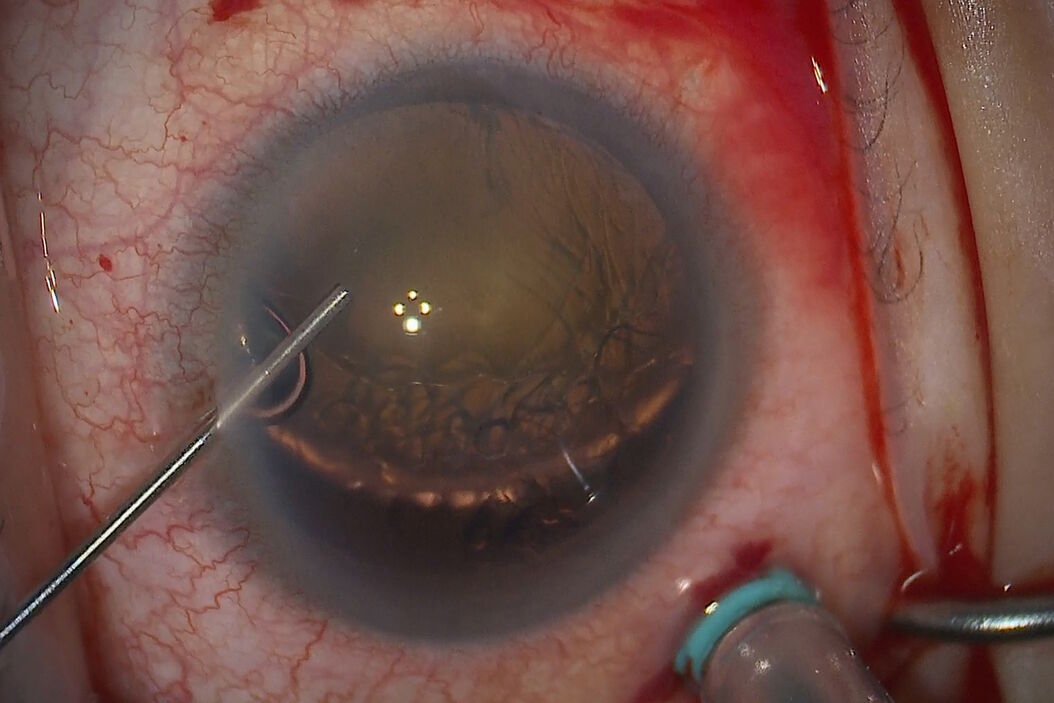
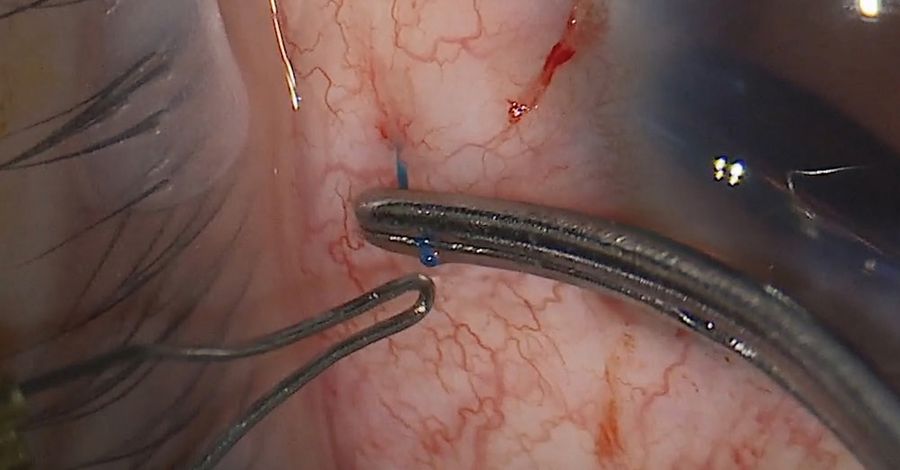
During phacoemulsification, a chop technique was used, and the quadrants were removed. After the hydrodissection, the posterior capsule reaction was assessed. As some of the lens material was removed, the lens was debulked with great care. A bed of epi-nucleus was left to keep the posterior capsule back. After the nucleus removal, the epi-nucleus was taken out removing the retractors. Retractors were removed with a technique of sliding the sleeve of the retractor out, then twisting it, essentially making it upside down to avoid the endothelium (Figure 2). The bag was then removed to do a Yamane lens fixation. A 3-piece hydrophobic acrylic intraocular lens was used, with a low refraction index and haptics made of polyvinylidene fluoride.
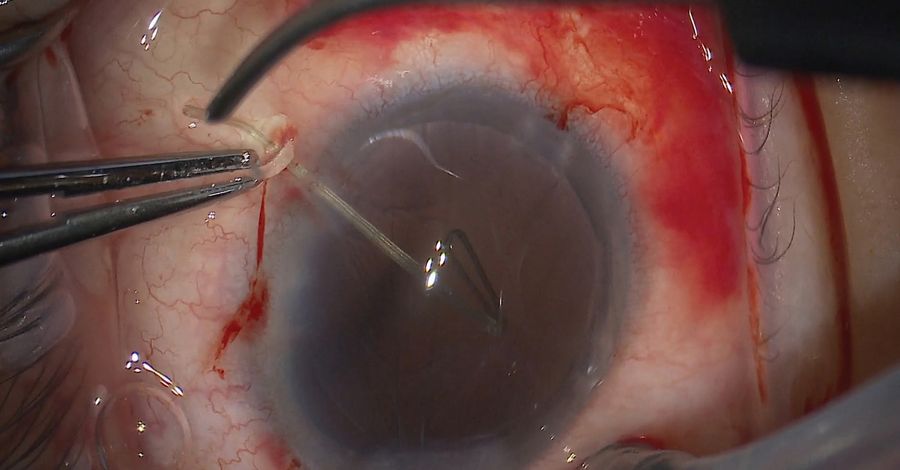
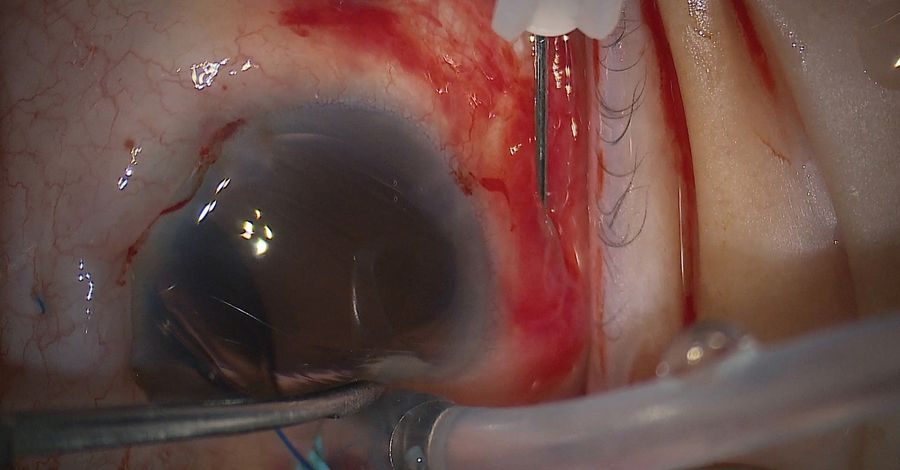
Tunneling was performed using a 27-gauge hypodermic needle to dock the initial haptic on the injector and avoid dropping the lens in the back of the eye (Figure 3). The injector was twisted by the assistant after the lens was loaded. Once the haptic was docked into the needle, it was withdrawn out of the sclera. As it came out of the sclera, the second haptic was left outside of the eye. Tires were then used to catch the haptic as it came out and avoid overpulling it.
While the haptics are very forgiving, there's still a chance of bending or pulling them out of the lens. Low temperature cautery was used to slightly bulb the haptic to prevent it from sliding back into the sclera (Figure 4).
The light reflex of the pupil was examined with the eye in an ortho position. The needle was then directed in the opposite direction to capture the trailing haptic (Figure 5). The corner apex was slightly nasalized. Using the light reflex instead of a toric marker for visualization, a micrograsper was used as close as possible to the
main wound, and the haptic was docked inside the needle.
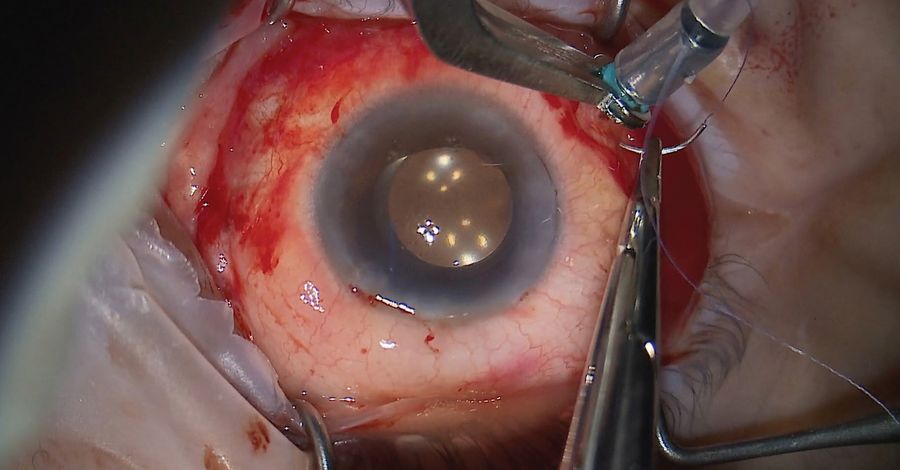
Around a third of the haptic was inside the needle. It was captured when coming out with the needle and cautery was again used to bulb it. It was then tucked back in the sclera. A scleral depressor was used to check that the haptic was flush inside the scleral wall.
Miochol-E was placed in the superior portion before using a low-cut setting and the forceps to draw the iris and make a peripheral PI. The surgery ended with the closure of the vitrectomy ports (Figure 6).
Post-operative outcomes
The surgery successfully treated the mechanism for the glaucoma, which was angle closure due to a dislocated cataract. The patient’s vision now corrects to 20/20. He no longer takes medication and his IOP returned to a normal range of 17mmHg.
Conclusions
The Proveo 8 surgical microscope from Leica Microsystems supports the specific needs of cataract surgery. It allows optimal visualization of all details throughout the procedure including the rhexis, the vitreous and the anterior chamber. It also provides optimal illumination and stable red reflex. The fine details can be picked out even with relatively low light, which is important to avoid macular toxicity.
Disclaimer: Please note that off-label uses of products may be discussed. Consult with regulatory affairs for cleared indications for use in your region. The statements of the healthcare professionals included in this presentation reflect only their opinion and personal experience. They do not necessarily reflect the opinion of any institution with whom they are affiliated or Leica Microsystems.