Morphology-based identification of cancer cells
From the early days, microscopy has helped illuminate how cancerous cells differ in size and shape from normal cells, providing insight into the underlying pathophysiology of cancer and offering ways to identify and diagnose cancers. Morphology-based pathology is still considered the gold standard for identifying and distinguishing cancer types [3].
The usefulness of morphological and histological techniques is not limited to clinical settings and is still heavily used in cancer research.
High-resolution imaging and fluorescence microscopy
While traditional brightfield imaging is still widely used in both clinical and research settings, the desire to understand the molecular mechanisms underlying cancer initiation and progression requires imaging techniques capable of visualizing proteins and molecules within cells. Fluorescent imaging offers a way to do this. It allows an understanding of the intracellular distribution of proteins and other molecules and how these change in cancer.
Fluorescent imaging of proteins and molecules can be achieved in multiple ways:
- Tagging with fluorescent proteins such as green fluorescent protein (GFP)
- Protein self labelling with HaloTag®, SNAP-tag® and CLIP-tag™
- Using fluorescently tagged antibodies that bind to targets of interest
- Direct fluorescent labels (e.g., DAPI, FITC, Alexa Fluor®)
Out-of-focus light impacts the resolution of fluorescent imaging, adding blur to images and making it more challenging to distinguish molecules and proteins of interest. Luckily, a wealth of imaging techniques have been developed that overcome this issue.
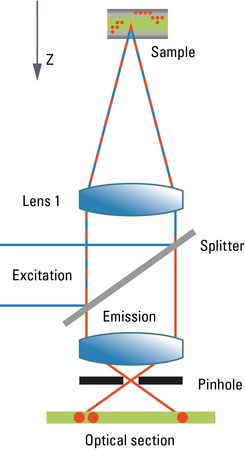
Confocal microscopy blocks out-of-focus light with the introduction of a pinhole, thereby achieving optical sectioning through the specimen under observation (Figure 1). Complete high-resolution images are created by moving this spot across the entire sample, and 3D images can be reconstructed from the high-resolution image stacks allowing us to visualize processes in three dimensions [4].
Schematic diagram of confocal scanning microscope, showing how the pinhole reduces out-of-focus light. The excitation light (blue) is coupled into the microscope by a splitter for incident illumination and focused to a diffraction-limited spot by the objective lens 1. Emission passes the splitter and is filtered spatially by the pinhole. Scanning the spot in x and y direction generates an image, the optical section.
Live cell imaging in cancer research
The need to preserve clinical samples to prevent physiological and molecular changes is critical since it is not always easy or possible to collect further samples from a patient. Such static, fixed samples can provide a wealth of information. Fixed samples, such as formalin-fixed paraffin-embedded (FFPE) tissues, allow researchers to study relevant samples.
The information available from fixed samples has limitations, the most notable being that they only provide a single snapshot in time. This restricts the information we can gain about dynamic cellular processes, including metastasis and cell migration [5].
Live cell imaging enables researchers to visualize such processes in real time, providing a greater understanding of them and their role in cancer. Fluorescence and live cell imaging offer a potent combination that can help better understand cancer initiation, progression, and treatment.
Several imaging technologies can visualize and record dynamic cell processes in various sample types, from cultured cells to organoids and even whole organisms. To gain meaningful results, it is vital to maintain
cellular viability, meaning samples must be kept at appropriate temperature, humidity, and CO2 levels.
Depending on the research needs, multiple technologies can be used to reliably and reproducibly image live cells, including widefield microscopy, confocal microscopy, light-sheet microscopy, multiphoton microscopy, and stimulated emission depletion (STED) microscopy.
The Mica Microhub from Leica Microsystems combines widefield fluorescence, confocal, and transmitted light microscopy, enabling reliable, high-resolution imaging of live cells. The advanced incubation chamber allows users to better mimic physiological conditions by regulating temperature, humidity, and CO2, ensuring cells remain viable, and data is more relevant.