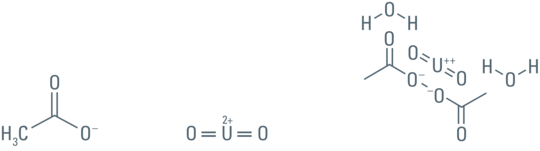Introduction
Therefore, the double contrast method of ultrathin sections on grids with uranyl acetate and lead citrate is the standard routine contrasting technique for electron microscopy. Although this technique is well known contrasting is one of the most critical steps of specimen preparation for electron microscopy. Besides manual handling, especially the properties of used chemicals, which can cause precipitation artefact, are the error factors.
The uranyl acetate (UA), which enhances the contrast by interaction with lipids and proteins, forms a yellow, needle-like crystal precipitate if not used in the right concentration and if redundant UA is not removed from the section. The lead citrate, which enhances the contrast by interacting with proteins and glycogens, will form a waterinsoluble toxic white precipitate (lead carbonate), if not used strictly under CO2-free conditions. Avoiding CO2 in a manual process is difficult for which a special procedure is needed.
The scientific demand of reproducibility, high sample quality with low reagent consumption on one side and the request of safe working conditions by avoiding contact with hazardous reagents on the other side can be satisfied with a standardised, closed and automated process.
According to these demands, Leica Microsystems developed the fully automated Leica EM AC20. Automated contrasting with ready-made pre-packaged UA and lead citrate stains ensures minimum user contact with reagents and eliminates nearly all traditional problems of manual staining such as uranyl or lead precipitates, health risks and high reagent consumption.
Stains
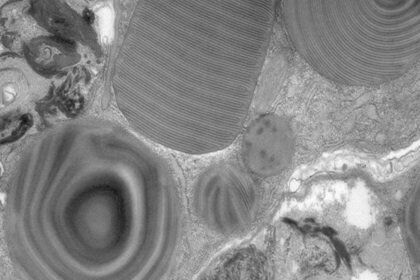
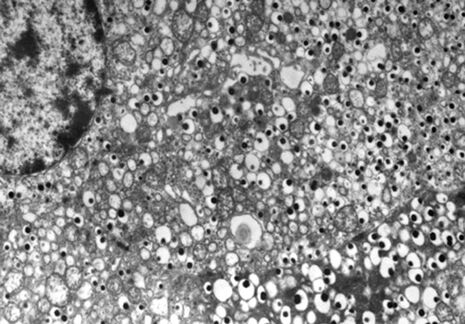
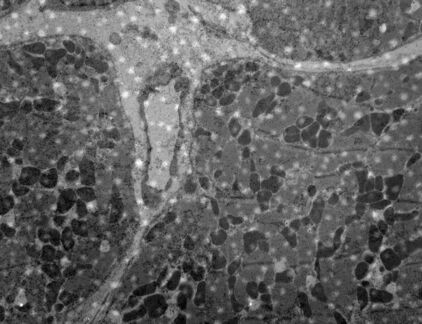
The most widely used stains in electron microscopy are the heavy metals, uranium and lead. The double contrast method of ultrathin sections with uranyl acetate (UA) and lead citrate is the standard contrasting technique for electron microscopy (Figure 1 and 2).
In this chapter we will discuss the characteristics of UA and lead citrate with their advantages and disadvantages depending on the contrasting process.
Uranyl acetate
The advantage of UA (Figure 3) is that it produces the highest electron density and image contrast as well as imparting a fine grain to the image due to the atomic weight of 238 of uranium. The uranyl ions bind to proteins and lipids with sialic acid carboxyl groups such as glycoproteins and ganglioside and to nucleic acid phosphate groups of DNA and RNA.
Gangliosides are glycolipids and are concentrated on cell surfaces. Glycoproteins are abundant in all membranes and part of the glycocalyx. Therefore UA delivers good contrasting results of membranes, nucleic acids and nucleic acids containing protein complexes such as ribosomes.
The disadvantage of UA is its sensitivity to light, especially UV, and will precipitate if exposed. It also precipitates when ageing. In addition, UA is both radioactive and toxic. Normal commercial stocks prepared from depleted uranium have a typical radioactivity of 0.37–0.51 µCi/g. This is a very mild level of radioactivity and is not sufficient to be harmful while the material remains external to the body. If ingested, inhaled as dust or by skin contact if skin is cut or abraded it is very toxic. The toxicity is due to the combined effect of chemical toxicity and mild radioactivity and there is a danger of cumulative effects from long term exposure.
Uranyl acetate solutions
UA is used either as an alcoholic (ethanolic or methanolic) or an aqueous solution. Both types of uranyl solution have individual advantages and drawbacks.
Alcoholic uranyl acetate
The advantage of an alcoholic stain is that it will penetrate more easily into the plastic embedded tissue and thus give a high contrast requiring only a short staining time.
Alcoholic solutions are prepared usually as saturated solutions in 50–70 % ethanol or in dry methanol.
The highest solubility is obtained in methanol. Consequently, the highest possible contrast is given by saturated methanolic uranyl acetate stain.
Alcoholic, especially, methanolic uranyl acetate is a chemically aggressive solution. Therefore, an enhanced extraction of cellular materials should be taken into consideration when using methanolic uranyl acetate. The alcoholic UA dissolves the colloidin-parlodion support film and can solubilize even formvar film if staining time is extended. It also has a disruptive effect on many plastics and rubbers.
Aqueous uranyl acetate
In order to obtain a specific effect of UA stain, it should be used as a water solution.
When UA is dissolved in water, the pH of the solution will range from 4.2 to 4.9 depending on the concentration (0.5 % to 3 %). At this pH the uranyl species with positive charge dominate and the stain solution gives an adequate contrast. Negatively charged molecules such as nucleic acids, stain particularly well.
The aqueous stain is labile and also very photo-sensitive in this pH-range. Aging of the solution and irradiation with light leads to precipitation, both of uranium hydroxide and of tetravalent uranium hydroxide.
Tetravalent uranium also precipitates in the presence of certain organic ions such as the unpolymerized monomers of the embedding medium as seen below:
Because stain has a low pH it is not recommended for use with specimens that are unstable in acid conditions. Also, the stain precipitates at physiological pH and in the presence of many salts and great care is needed when using it. Therefore, the filtered stain should be stored in the dark at 4 °C and can be used for several months.
Stabilized solutions
Stabilization of UA is required to avoid precipitation for longer storage times. The precipitation can be completely avoided when UA is stabilized by acidification. The acidification however, considerably decreases the staining effect. At pH 3.5 the attachment of uranyl ions to DNA is strong and also more specific than at pH 4.0 since at this pH only DNA has the negative charge. Below pH 3.5 uranyl stain will react very weakly with the tissue components since most proteins and even the nucleic acids are no longer negatively charged at lower pH values.
Leica Ultrostain I (Uranyl Acetate Solution)
The Leica Ultrostain I corresponds to a stabilized 0.5 % uranyl acetate solution with a pH of 4.4. It is pre packed in brown 200 ml bottle to protect it from light. This is done at a chemical production plant in a special risk laboratory, under conditions which are hardly possible to arrange in an EM-laboratory.
Precipitate
In the routine contrasting procedure, the problems with UA are relatively easy to master by a repeated renewal of the stain and by daily filtering or centrifugation. The UA crystallizes in needles in large squares or rhombi-formed crystals (or as granular aggregates, Figure 4).
Preparation of 4 % (w/v) stock solution of aqueous uranyl acetate
Caution: Uranyl acetate is extremely toxic due to the combined effect of chemical toxicity and mild radioactivity and there is a danger of cumulative effects from long term exposure. Exercise care in weighing the powder, always wear latex gloves, lab coat, protective mask and other suitable protection (contact lab safety officer!)
Materials
- Latex gloves, lab coat, protective mask
- 200 ml brown stock bottle (scrupulously clean!)
- 100 ml volumetric flask (scrupulously clean!)
- 50 ml pipette (scrupulously clean!)
- Balance under fume hood
- Weighing dish
- Hot plate
- Magnetic Stirrer and stir bar
- Whatman #1 filter paper
- Glass funnel
- Uranyl acetate dihydrate powder (depleted) [U(C2H3O3)2 × 2H2O], 4 g
- CO2-free double-distilled water, 125 ml
Note: Once glassware is contaminated with heavy metal stains it should not be used to prepare other chemical solutions.
Procedure
- Weigh 4 g of uranyl acetate (UA) under the fume hood (wear protection!) and add it to the 100 ml volumetric flask. Pipette 96 ml of near-boiling CO2-free double-distilled water into the flask with the UA.
- Place on stirrer until uranyl acetate dihydrate crystals are dissolved. This will take a while. After dissolving let the solution cool down to room temperature.
- Filter the UA through the Whatman #1 filter into the 200 ml brown bottle (light protection) and cap tightly and label. This stock solution can be stored for months at 4 °C.
- Rinse contaminated glassware into a waste bottle for UA solution and store separately from laboratory glassware. Solid waste that is contaminated with UA should be disposed of in a solid waste container set aside for radioactive waste (contact lab safety officer!).
Lead citrate
Lead citrate enhances the contrasting effect for a wide range of cellular structures such as ribosomes, lipid membranes, cytoskleleton and other compartments of the cytoplasm. The enhancement of the contrasting effect depends on the interaction with reduced osmium, since it allows the attachment of lead ions to the polar groups of molecules. Osmium is used routinely as a fixative. Lead citrate also interacts, to a weaker extent, with UA and therefore lead citrate staining is employed after UA staining.
The disadvantage of lead citrate is its unpredictable behaviour under laboratory conditions and the time consuming and delicate preparation (see Figure 7). Lead citrate precipitates easily in the presence of carbon dioxide, either in air or in the water used for preparing the stain or for rinsing. Lead carbonate (PbCO3) is a water insoluble toxic white precipitate and accordingly will not dissolve in rinsing water. In general all lead salts such as lead citrate are extremely toxic.
Lead solutions
The staining effect of lead solutions is highly dependent, like the uranyl solutions, on the pH of the solution. Unlike uranyl, however, it is possible to use lead based staining solutions at very high pH values (>12 pH).
The alkaline Wenable & Goggeshalls and Reynold’s lead solutions are the most popular. From these methods, that of Reynolds (1963) is more reliable for routine work, simply because the commercial quality of lead citrate does not always correspond to the quality required for staining. In the Reynolds method, lead citrate is formed as a product from the reaction between sodium citrate and lead nitrate.
All alkaline lead stains can be considered to be stabilized solutions. Therefore, those solutions are suitable for commercial handling, provided they are made under conditions which eliminate the formation of lead carbonate.
Leica Ultrostain II (Lead Citrate Solution)
The Leica Ultrostain II corresponds to Reynolds lead citrate. The 3 % lead citrate solution is pre packed in vacuum tight bags under helium atmosphere. This is done at the chemical production plant in a special risk laboratory under the utmost care, and in conditions which are not possible to achieve in a standard EM-laboratory.
Precipitate
Lead carbonate (PbCO3) is a water insoluble toxic white precipitate.
It appears as black grains in the electron microscope. The grains can either be large (Figure 7a) and few or the section can be covered with a fine deposit (Figure 7b).
Materials
- 50 ml volumetric flask with plastic or glass stopper (scrupulously clean!)
- 25 ml pipette (scrupulously clean!)
- pH-Meter (scrupulously clean!)
- Magnetic stirrer and stir bar
- Lead nitrate [Pb(NO3)2], 1.33 g
- Sodium citrate [Na3(C6H5O7) × 2H2O], 1.76 g
- CO2-free double-distilled water, 100 ml (freshly made and chilled to at least 25 °C)
- 1 N NaOH, 10 ml (freshly prepared in CO2-free double-distilled water)
Procedure
- Combine 1.33 g lead nitrate, 1.76 g sodium citrate and 30 ml CO2-free double-distilled water in the 50 ml volumetric flask. Shake the solution vigorously for several minutes then 5–6 times over a 30 min period. This will generate a milky white suspension of lead stain with no large particles. If one sees large chunks, continue shaking until the particles are dissociated, or start over. Stir on stir plate until uranyl acetate dihydrate crystals (Figure 3) are dissolved. This will take a while. After dissolving let the solution cool down to room temperature.
- Add 5 to 7 ml of 1 N NaOH to the suspension while stirring. The milky solution should turn clear. If not, add a few more drops of NaOH up to total 8 ml. If the solution still does not turn clear, something is wrong and the stain should be discarded! Rinse contaminated glassware into a waste bottle for UA solution and store separate from laboratory glassware. Solid waste that is contaminated with UA should be disposed of in a solid waste container set aside for radioactive wastes (contact lab safety officer!).
- Withdraw a small amount of clear stain and check the pH using a pH meter. The pH should be 12.0 +/- 0.1. If the pH is too low add more NaOH to the clear solution in the flask of step 2. If the pH is above 12.1 start over.
- After pH has been verified, add CO2-free water to the volumetric flask to bring the solution to a final volume of 50 ml. The flask should be tightly stoppered.
- The solution has a shelf life up to 6 months if sealed tightly. Before use, centrifuge (5,000 × g/10 min) or passage through a microfilter.
Note: An exact pH is extremely important with this stain. If the pH varies by more than 0.1 unit from pH 12.0, poor staining or precipitation will occur.
Contrasting procedure
Manual contrasting
In the preparation of specimens for electron microscopy, the manual staining of sections is the step in which most failures are experienced. Here, the grids are placed on drops of uranyl acetate (UA) solution and removed after a certain incubation time with forceps (Figures 8a and 8b). Then the grids are rinsed in water several times before being placed on drops of lead citrate for incubation (Figures 9a and 9b). In order to prevent precipitation of the lead citrate by exposure to CO2, pellets of NaOH are added to the staining dish (here a Petri dish) to exclude atmospheric CO2. In addition, the rinse water should be CO2-free (double distilled). After staining, the grids are rinsed again in several water baths and by using a jet of water (Figure 9c). After wicking and drying, grids can be analysed.
The major problem of the manual staining procedure is the precipitation of the metallic salts during the staining process, especially the precipitation of lead. The usage of NaOH pellets to minimize the precipitation is a commonly used technique. Even with the best precautions, precipitation may destroy many hours or even days of work, during the critical half-hour of staining.
Another widely experienced problem is the repeated handling of grids which can easily damage the support films and even the sections.
Since many steps of manual handling are included, the presence of the user is necessary during the whole staining time. Also, when only a small number of grids can be stained simultaneously, the procedure must often be repeated. Therefore, manual staining contributes greatly to the work load in the EM laboratory. If multiple sequence or the staining of long serial sections is to be performed, and especially if short staining times are used, consistent reproducibility is difficult and sometimes impossible to obtain.
Finally the increased health risk by repeated handling of the dangerous chemicals during the preparation of stains and staining is a main disadvantage of the manual staining.
Automated contrasting
The Leica EM AC20 automates the traditional manual technique of double-contrasting of ultrathin sections mounted on grids. The technical design offers a solution to the majority of the traditional problems of manual staining such as precipitation and unequal staining results. Besides, by contrasting up to twenty grids simultaneously, the automation speeds up laboratory routines and standardises the contrasting result. Therefore, fully automated staining with the EM AC20 gives a widely appreciated improvement to the electron microscopy laboratory routine by eliminating the traditional staining problems, reducing the health risks and giving the technician time for more rewarding work.
The instrument uses pre-packed, stabilised solutions of uranyl acetate (UA) and lead citrate to minimise the user's contact with hazardous chemicals and eliminates precipitation and contamination due to controlled and standardised stain preparation and bottling (see chapter 2). The heart of the Leica EM AC20 is the peristaltic pump and non-contact valves system, which means the reagents travel through nothing but the air-sealed chamber tubing. This pump-valve-tubing system contains two independent tubing set-ups. One allows controlled streaming of reagents and rinse to the grid chamber (Figure 9b). The second guides the waste from the chamber to the specific waste bottles.
Factors affecting the contrast
In the staining technique, the factors with the highest impact on contrast quality are the quality and concentration of stain solutions. Beside the stains, the factors such as fixatives, buffers, rinse water, dehydration, rinse time, type of embedding medium and the level of polymerization as well as the section thickness influences the contrast too.
General factors
Fixatives
Osmium tetroxide fixative enhances the contrast. It acts as fixative as well as enhancer of contrast during post-staining by interacting with uranyl acetate and lead citrate.It has been indicated that fixation time has an effect on contrast obtained by uranyl acetate contrasting. A long fixation with Osmium tetroxide decreases e.g. the contrast of chromatin.
Buffers
Uranyl reacts strongly with cacodylate and phosphate ions which can lead to uranyl salt precipitation. Therefore a higher contrast without precipitation is obtained when washing the samples after fixation extensively.
Phosphate buffer can remain in the tissue even after dehydration and embedding and often gives rise to small, local, dense deposits in muscle and nervous tissue.
Rinse water
The CO2 content of the rinse water affects the contrast by forming lead carbonate precipitation. Fresh distilled water has pH close to neutral. If distilled water is allowed to stand for a long period of time, CO2 is dissolved from the air.
In addition, a low pH <6.0 of the rinse water decreases the contrast given by uranyl acetate and therefore also the general contrast. If the pH of water is lower than 6.0, KOH should be added for neutralization. The rinse water should also be degassed or boiled to prevent the formation of air bubbles which, if present, attach onto the grids in the staining chamber and prohibit staining.
Embedding medium
Epon and Epon substitutes have been found to contrast in similar ways and therefore do not need special handling.Araldite is known to give an inherent background stain. Shorter contrasting times at lower temperature are therefore recommended.Spurr embedding resin generally requires a longer contrasting time. It is advisable not to increase the temperature when staining Spurr, since like Araldite, the resin background itself has a tendency to take up the stain.The age of the embedded specimen is important, since polymerisation of the embedding material continues during storage. Old specimens and sections require longer Uranyl Acetate contrasting time.
Section thickness
Thin sections show lower contrast than thicker sections but higher resolution, since there are fewer sites for the stains to be bound to. Therefore thin sections require longer staining times than thicker ones.
Level of polymerization
A poorly polymerised embedding medium is soft, difficult to cut and takes up stain rapidly. Therefore a shorter contrasting time at lower temperature is recommended.
En bloc staining
Contrasting of the specimen before embedding has become popular in recent years as a method of enhancing contrast without precipitate formation.
However, "en bloc" staining alone does not enhance the contrast of the specimen sufficiently for most studies. Post staining of the sections with both, uranyl acetate and lead citrate is recommended in most text books.