FluoSync - simultaneous multilabel imaging
Find out how to detect multiple labels at the same time, without worrying about crosstalk.
FluoSync is a streamlined approach for simultaneous multiplex fluorescent imaging using a single exposure
Traditional fluorescence imaging methods typically image each channel sequentially to reduce crosstalk between fluorophores. Multispectral imaging and subsequent linear or phasor-based unmixing have been independently described previously. Each required either tedious manual adjustments or in-depth understanding of the underlying technology or both. With FluoSync Leica Microsystems introduces an integrated approach eliminating the complexity while retaining the benefits of fast and gentle imaging. FluoSync is for the first time fully integrated into Mica, empowering the world's first Microhub. FluoSync captures photons across the visible spectrum, discarding less information than narrow bandwidth filters, and then separates each signal by employing a phasor-based Hybrid unmixing approach, which results in robust channel separation.
Multiplex Fluorescence imaging has never been easier
Multicolor widefield microscopy
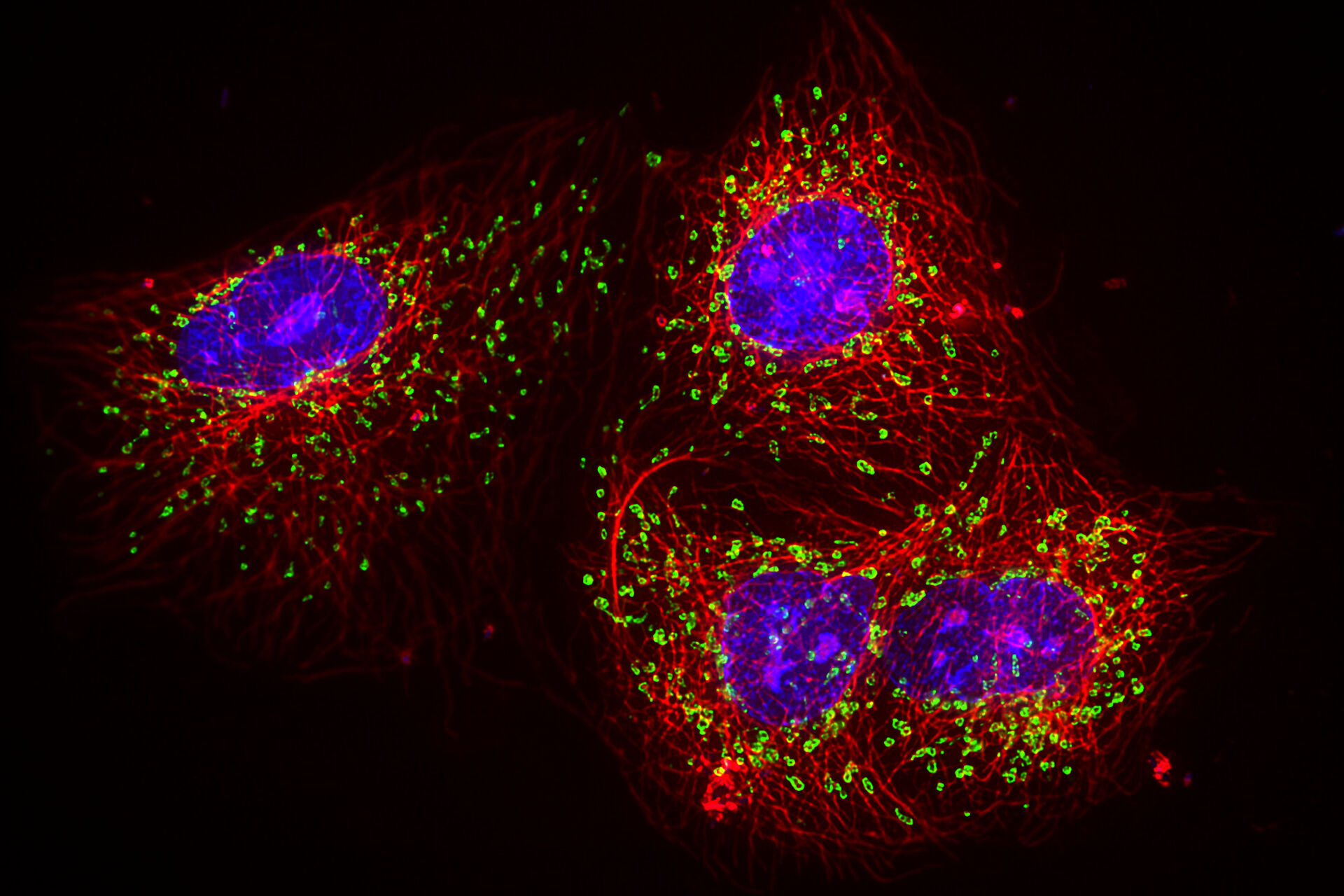
Fluorescence microscopy offers a powerful tool for studying the cellular function of proteins and other biomolecules. It has become an essential tool for life sciences research due to its unique ability to label and track target molecules with a high degree of specificity. Antibodies and genetic tags can be used to target and label molecules with spectrally resolvable fluorescent dyes (see Figure 2). Using a combination of these fluorescent tags enables users to track and study the behaviour and interactions between the distinct labelled populations of molecules within a specimen.
Standard approaches to obtain a multicolor fluorescence image need to consider the spectral overlap of the fluorophore signals, commonly known as crosstalk or bleed through as shown in figure 2b. Typically, researchers avoid crosstalk by keeping the number of dyes in the sample to a minimum (i.e. one or two dyes at most), as the more dyes used in a sample, the higher the chances of encountering issues with signal crosstalk. However, the increasing need of more biologically relevant data requires researchers to look at three or more events in a single sample, making this simplistic approach no longer an option. In a typical multiplex experiment, one or two fluorescent dyes are used to identify specific cellular structures - such as the nucleus or mitochondria - and additional dyes are employed to identify proteins of interest.
Spectral separation and unmixing
When multiplexing, dye combinations with good spectral separation are normally required. A good example is the combination of Alexa 488 and Alexa 555 as illustrated in Figure 2, which emit light in the green and red part of the spectrum respectively. But the spectral separation of dyes in a sample stained with multiple fluorophores is often not straightforward. The emission curves of dyes can partially overlap, resulting in part of the fluorescence signal emitted by one dye falling into the detection window of its spectral neighbour. In other words, some signal from your green dye bleeds through to your red channel. This problem can be worse in live cell imaging, as users try to avoid the blue end of the spectrum to avoid phototoxicity. Researchers still need to use multiple dyes to tag proteins of interest, but are limited to a narrower region of the spectrum, increasing their potential crosstalk issues.
There are two common methods to spectrally unmix fluorescent signals: Separation with specific narrow band-pass optical filters and linear unmixing, also known as multi or hyperspectral imaging.
Channel Separation with specific narrow-band optical filters
The simplest approach to multiplex is to image the dyes sequentially. This way, each dye in the sample is excited and its emission detected separately. Excitation wavelengths are controlled using excitation filters or wavelength specific LEDs. Optical band-pass filters in filter cubes are moved into the light path between each image to check the spectra of the light reaching the sample and detector. The final composite multicolor image is produced by overlaying the separate exposures. Sequential excitation of dyes helps reducing crosstalk issues, without fully eliminating them. This approach might be too slow to image fast cellular events, such as vesicle tracking or calcium imaging. Often an object of interest will have moved between two frames, creating an artifact known as spatiotemporal mismatch. This makes it impossible to accurately track an object in two channels unless both channels are acquired simultaneously. Furthermore, when imaging large samples, like multi-well plates, the long acquisition cycle can significantly increase the run time for an experiment, reducing productivity.
To speed up the acquisition process, it is possible to use multi-band-pass filter cubes combined with fast filter wheels in what is known as Sedat and Pinkel configurations. These small filter wheels can be switched much faster than filter cubes. Multi-band-pass filter cubes block fluorescent emission from dyes outside its narrow emission windows, resulting in the potential loss of precious signal (see Figure 2a). This technique might also increase sample photobleaching and reduce temporal resolution and signal-to-noise levels in the final image.
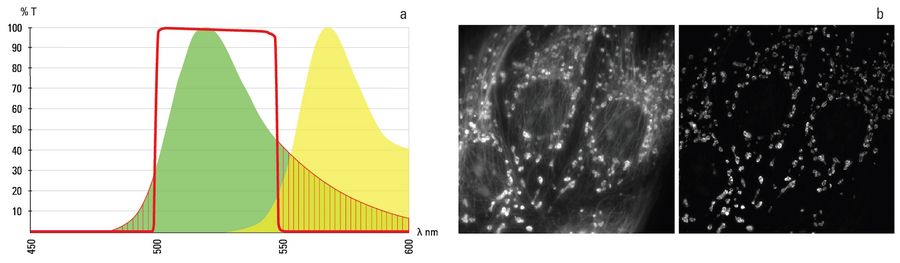
Figure 2: a) The fluorescence emission profile of Alexa 488 (green curve)and Alexa 555 (yellow curve). The overlap of the two emissions spectra is plain to see. The red line indicates the band pass of a 488 emissions filter. Only the emission wavelengths fitting under this line makes it past the filter to the detector. The red shaded areas show the part of the 488 signal (~20%) blocked by the filter to limit cross talk with adjacent dyes. b) The 2 images in Figure 1b show channel bleed through (image left) and its suppression (image right) by using a band pass filter. In the cases the red dye used to stain the micro tubules can clearly be seen in the image of mitochondria as the image was acquired using a multi band pass filter cube but without an emissions filter in front of the detector. The red dye (microtubules) is cross excited with the green dye (mitochondria) leading to sever channel bleed through. In the second image the cross talk has been eliminated by using the correct emissions filter.
Linear unmixing
Another approach to separate dyes is linear unmixing which uses mathematical unmixing methods to separate the channels. In this method, the microscope collects multiple images of the sample, each covering a different spectral band (see Figure 3). The data collected contains a spectral curve for every pixel with the combined emission spectra from all the fluorophores. Spectral linear unmixing is then used to quantify the contribution of each fluorophore population to the signal and a multichannel image is then created. In widefield imaging, this technique is known as multispectral imaging. In confocal imaging, where the system can often scan with higher spectral resolution, this approach is referred to as hyperspectral imaging or Lambda scanning. Unless you have a specialized hardware able to capture the multiple spectral windows simultaneously, this approach requires repetitively exposing the sample resulting in extreme light stress making it unsuitable for live cells. Linear unmixing requires a known reference spectrum for each dye in the sample. Ideally, the reference spectrum is acquired using the same type of sample and preparation method to enable the system to separate the dye signal from background noise and autofluorescence unique to the sample.
Linear unmixing is based on matching signal levels in calibration curves to the data acquired from a real sample. Variations in dye concentration, where signal from one channel is much brighter than another, can lead to unmixing errors. Additionally, the presence of detector or optical noise (sample background fluorescence) leads to the risk of introducing further sources of errors. These vulnerabilities can mean that linear unmixing is
unsuitable for low light levels, samples with high background noise, and samples with significant intensity variation between channels.
FluoSync
FluoSync is a fast and reliable method for unmixing signals from multiple fluorophores in a sample. It streamlines the approaches of multispectral imaging combined with phasor analysis (Cutrale et al. in 2017) and offers several advantages over more traditional methods for multiplex imaging.
FluoSync acquires multispectral image data by exciting and detecting all channels simultaneously with a single detector exposure. The detectors used for this approach employ multiple sensors, each acquiring a different part of the spectrum. The resulting combined dataset is then unmixed using Phasor-based Hybrid unmixing.
As all fluorophores are acquired simultaneously, it is faster than traditional methods, making it a suitable solution for live cell imaging and imaging of large samples, such as microtiter plates. Simultaneous dynamic live cell imaging benefits from the speed and high synchronicity of the image acquisition. Fast moving objects like vesicles will not have moved between capturing the signal from two different fluorophores. Imaging functional assays like e.g. the live dead assay on larger sample carriers such as microtiter plates will benefit from the speed advantage and allow to correlate the data with additional markers at no additional time spent for the acquisition. Unlike approaches which rely on multiple bandpass filter cubes that block part of the emitted signal to improve channel separation, FluoSync uses broad band filters collecting most of the emitted signal, providing an efficient detection solution also for low light imaging. The collected light is split via built-in spectral splitters. Hence, picking and installing the right filter cube is now a thing of the past.
Phasor-based spectral unmixing
Phasor-based analysis was originally conceived as a powerful method for visualization and rapid analysis of fluorescence lifetime imaging microscopy (FLIM) by Digman et al. in 2008. Later this method was adapted to spectral imaging by Fereidouni et al. in 2012 and Cutrale et al. in 2017.
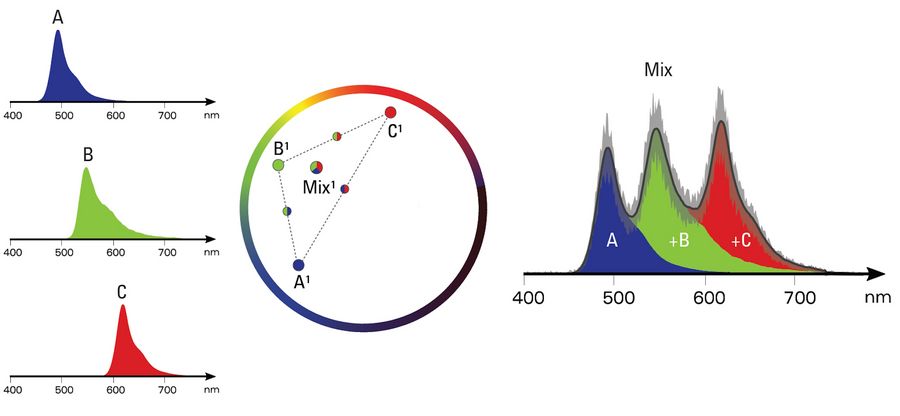
As a rule of thumb, the transformation into the phasor will place the spectrum on a radial color axis and the width of the spectrum defines how far away the spectrum is from the origin. Mixtures of two pure dyes will fall onto a straight line between the pure ones, while the distance to the pure dye represents the relative contribution to the overall signal. What holds true for any two dyes, extends to more dyes as well. Each possible combination of dyes will end up in a defined position in the phasor space.
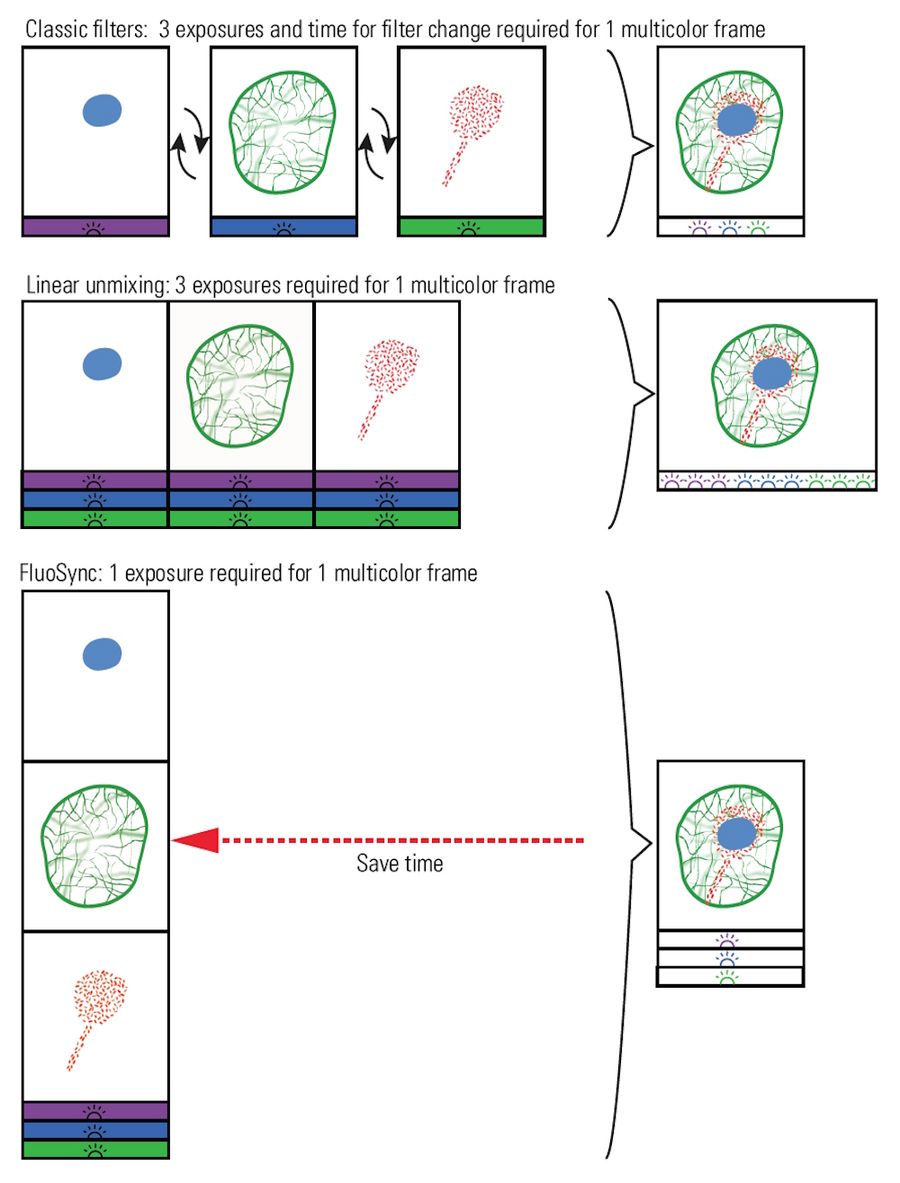
Figure 3: Overview of the three methods discussed to separate colors when imaging a sample with multiple fluorescent dyes. In the comparison, the advantage of improved acquisition speed compared to conventional fluorescence imaging approaches of FluoSync becomes apparent. FluoSync captures the full emitted spectrum from excited dyes, so photons are not wasted making it an ideal detection technology for low light imaging of sensitive living samples.
The phasor representation of spectral information allows for real-time, semi-blind spectral unmixing of the different fluorescent components in the image. The use of this representation is not only limited to fluorescent labels, but also allows unwanted signals, such as autofluorescence, to be eliminated, making it a more robust approach when compared to linear unmixing. The phasor-based unmxing is best at unmixing signals of up to 3 fluorophores. For more than 3 fluorophores, the phasor still allows to easily identify the strongest contributor.
Most recent developments in the field of unmixing spawned a hybrid unmixing - combining the strength of each (phasor-based and linear) unmixing (Chiang et al. in 2022).
Hybrid unmixing explained
FluoSync capitalizes the power of a hybrid between phasor-based and linear unmixing. Phasor analysis is used for rapid clustering of similar spectra and spectral denoising and linear unmixing for identification of the dye’s individual contributions for each cluster even beyond 3 dyes (Figure 4).
Figure 4: Depicted on the left are three individual spectra of a blue, green, and red fluorophore. By using the phasor analysis each pure spectrum will fall into a defined space in the phasor space where the color is represented on a circle and the sharpness of the signal determines the distance to the center (middle panel). Any combination of these fluorophores will also fall into a defined space. Depicted are one combination for each of the three fluorophores and a mix of all three. As possible combinations of fluorophores will also fall into "their" space, the spectra can be averaged for denoising. One example is shown in the right panel, where the black line represents the average ± the error (depicted as gray area). The noise-reduced spectrum represents a sum of all contribution fluorophores, that fills the area under the curve nicely.
Like the pure phasor-based unmixing, all pixels with similar fluorescence composition will fall into the same space in the phasor. Because of this fact they can be averaged to achieve spectral denoising, irrespective of their spatial position, i.e. the origin position in the image. This reduces the number of operations of linear unmixing by manifold: the number of pixels (typically in the megapixel range) to typically less than 100,000 operations. The contribution of every dye is then identified by applying linear unmixing to the noise-reduced spectra.
When compared to linear methods, the hybrid unmixing utilizes the power of phasor-based speed and averaging advantages without compromising on the amount of dyes that can be separated.
Summary and Benefits
FluoSync is the digital advancement of optical filters. It is a robust method which can be used to image multiple dyes. As it requires only a single image exposure and can be done with an automated spectral unmixing approach, it results in easier and faster multicolor fluorescent imaging.
Compared to traditional imaging techniques, FluoSync offers high speed, simultaneous multi-channel acquisition, while making use of crosstalk instead of avoiding it. It is an ideal solution for scanning large samples or capturing fast dynamic processes in live cells. Finally, FluoSync simplifies the experimental workflow by eliminating the need to manage multiple sets of filter cubes between experiments. So you can focus on getting results instead of understanding your microscope.
FluoSync in Mica
Mica is the first product designed around the FluoSync detection concept to yield an easy to use, yet highly versatile microhub. Empowered by FluoSync, Mica brings you rapidly from sample overview to high resolution images, allowing you to effortlessly capture quad channel data sets without spatiotemporal mismatch in a fraction of the time.