How was your experience working with STELLARIS 5?

Dr. Timo Zimmermann: My experience using the STELLARIS 5 system was very positive. Getting started to use the system is very straightforward and I could easily apply my existing knowledge about confocal imaging when setting up an experiment. I was particularly pleased with seeing how confocal imaging is now extended to include lifetime-based information. I liked how STELLARIS makes lifetime-based information accessible using the different TauSense tools1, something that was quite complex before. Now, with a click of a button, fluorescence lifetime measurement becomes just another additional feature of your confocal experiment and the possibilities of having this extra contrast are very interesting for me.
In addition, STELLARIS appears to be very light efficient thanks to its new detectors that work both in analogue and photon counting modes: using very little power, you get excellent signal outcomes.
Tell us more about your experience using TauSense.
Dr. Timo Zimmermann: I used a lot the TauContrast feature that adds the average lifetime information of every pixel as a separate image channel. Because you can use pulsed illumination as standard excitation over most of the spectrum, you actually get the lifetime information for free with the detectors. This is really important, because it gives you another dimension for each fluorescence signal, actually one of the defining characteristics of a fluorophore, in an extremely straightforward manner.
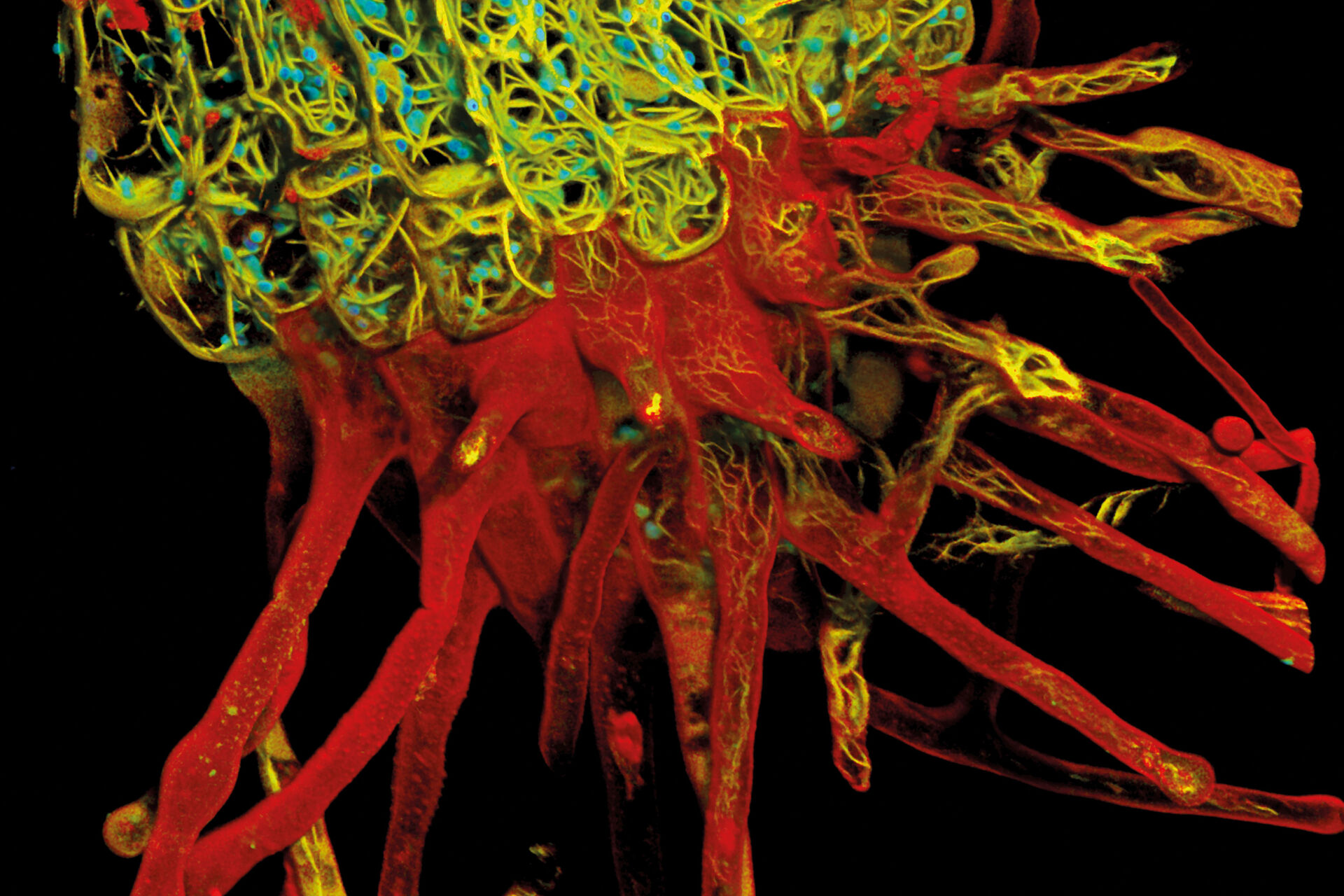
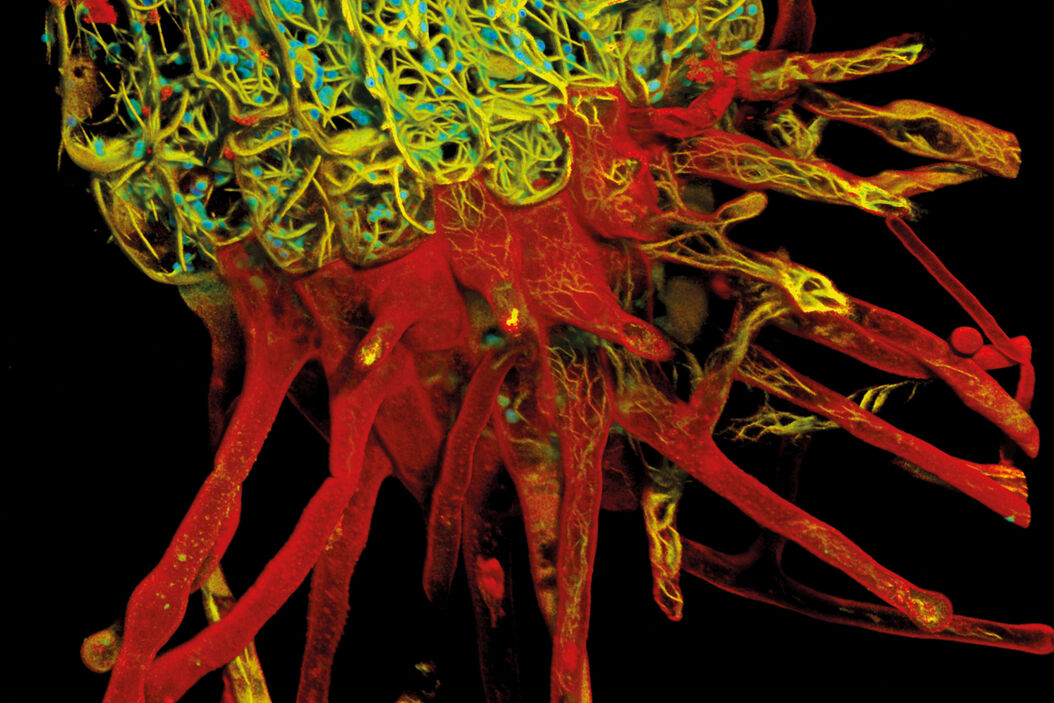
I used TauContrast mainly for a fairly complex biological sample. Here, the lifetime information added a lot in addition to the less specific spectral information, so you could distinguish the structures much better. In other words, it helped in understanding the complex morphology significantly better. I believe that for visually and structurally complex samples with a lot of natural information, not just the fluorophores you add, it is very useful, as you can see these structures in addition to the labelled structures.
So, using TauContrast gave you more information or did it allow you to separate the information that you had?
Dr. Timo Zimmermann: It gave me more information. If the only fluorescence signals in the sample are the ones you introduce with established fluorescence labels and these are well distributed over the spectrum, it is not needed. But when you have more information, such as a sample with background or a complex sample, using lifetime information helps separate different pieces of information, so it is much easier to understand when there are different structures that just look similar in their emission spectrum. A fluorophore is characterized by its excitation and emission spectrum and its lifetime. Getting all three literally gives you the whole picture. Given the significant overlaps that can exist in fluorescence spectra, this really helps.
What would be other uses of lifetime information that researchers could take advantage of?
Dr. Timo Zimmermann: Background removal in thicker samples using TauGating is very useful. This is one of the features that you can use to gate your signal into different areas and, with those gates, remove the background. For demanding samples, such as thick samples or oversized embryos, having lifetime information on the side has a lot of potential, because it would allow you to focus on what’s important and helps you understand these complex samples better. Samples like these – think organoids and model organisms - are becoming more important as they resemble real biology much better. And you can observe these samples much better with new technology like TauSense.
Having fluorescence lifetime information on the side helps you understand complex samples better.
Dr. Timo Zimmermann
Besides access to lifetime information, what other benefits do you think STELLARIS can offer to core facility users?
Dr. Timo Zimmermann: It’s a very complete system, as it also has the unique features of its predecessor, such as the resonant scanner, the AOTF (acousto-optical tunable filter) and AOBS (acousto-optical beam splitter) that enable the removal of all glass filter and beam splitter components, or spectral freedom thanks to the white light laser. For me, these still provide key benefits for confocal experiments and STELLARIS improves these features and adds to them.
If you are running a core facility, your system has to be flexible and this is reflected in the design of the system. STELLARIS can do everything we need a confocal to do at a core imaging facility, especially as it has no limitations on the detection side and you have the freedom of the white light laser and can further adapt the excitation side as needed, adding solid-state lasers if you need a lot of power. And, as mentioned before, the fact that the white light laser is pulsed and that, thanks to it, you can use lifetime information basically for free, is a huge advantage.
So, it’s just a very flexible system. It provides freedom for confocal experiments from all different angles: the resonant scanner provides speed when it is needed as it can be switched in and out during the imaging session and there are also the very good all-purpose Power HyD detectors that can change between analog and photon counting modes. Having both modes is a really nice feature that provides you with many possibilities. You can easily switch between modes and also set them separately for each detector so that you have a lot of choice on dynamic range. The software interface, ImageCompass, has a new design for fluorescent channel selection that provides good accessibility. So, for me, STELLARIS has an all-around applicability for confocal imaging.
The interview was held in February 2021, after Timo Zimmermann had returned to the EMBL in Heidelberg.