A brief history
In the 1893 paper entitled "Ein neues Beleuchtungsverfahren für mikrophotographische Zwecke" which was published in Zeitschrift für wissenschaftliche Mikroskopie und für mikroskopische Technik (“A New System of Illumination for Photomicrographic Purposes” in the Journal of Scientific Microscopy and Microscopic Technique) he set out the main problems microscopists faced at the time. Auer gas lights and zirconia light were the main sources of illumination available in this era, but these had the tendency to produce an unwanted ‘glare’.
The Auer gas light was named after the Austrian scientist and inventor, Baron Carl Auer von Welsbach (1858-1929). He is credited for patenting the gas mantle which was used as street lighting throughout Europe. Although Auer gas lights still produced carbonic acid and a certain amount of heat, the London Journal of Gas Lighting and Sanitary Water Supply exclaimed that the Auer light was “eminently suitable” for microscopy.
The alternative zirconia light was an intense chemical light source produced by heating the element zirconium. However, with such light sources, Koehler declared that “it is difficult or impossible to achieve uniform illuminations”. Koehler realised that for consistency in illumination, every single component of the microscope needs to be considered and correctly aligned in relation to each other. By making adjustments to the diaphragms and condensers, he overcame the main problems of overheating and light consistency (even using an opera glass in place of a convex lens!).
An important principle
Even today, his consistent illumination technique is still widely used and forms the basis of many techniques including confocal, phase contrast and differential interference contrast (DIC) microscopy. Once you know how to set up a microscope for correct Koehler illumination, it should become second nature and will transform your imaging work. It is simply one of the most important principles of light microscopy.
It takes less than a minute to set up a microscope for Koehler illumination. This will ensure that the light source evenly illuminates your specimen, produces a sharper image and will increase the contrast between areas of staining or between regions of tissue in your sections. You will also be able to collect more imaging data from each of your specimens.
If you require images for use in Powerpoint presentations, poster presentations or for submitting to journals, then Koehler illumination is a must. This simple five step technique goes a long way and, with a little practice, will easily become second nature before you capture your images.
As well as the microscope objectives, there are two other components which you should be aware of (and which are adjusted) for Koehler illumination.
The field diaphragm
The ‘field diaphragm’ is usually located in the base (in an upright microscope) of the instrument as part of the microscope body (Figure 1). Some older (or simpler) microscopes may just have an ‘on/off’ switch for the light source and there may be no field diaphragm to control the intensity of light from the source. The field diaphragm simply controls the amount of light which finally reaches the slide on the stage. By controlling the amount of light coming from the light source, you are able to reduce glare from the source and this results in images with greater contrast. The field diaphragm is usually controlled by a knurled ring around the lens.
The sub-stage condenser
Directly under the stage in an upright microscope (or above the stage but in front of the light source/field diaphragm in an inverted microscope), is the sub-stage condenser (Figure 1). Although it has focus wheels which are similar to the fine/coarse focus of the microscope, this component is used for focusing the light from the light source/field diaphragm and not to bring your specimen into focus.
The ‘sub-stage condenser’ collects the majority of the light from the source and focuses it as a cone of image-forming light on the slide placed on the microscope stage. When this condenser is correctly adjusted, the light which illuminates your specimen and enters the objective is fully optimised. This results in a uniform intensity and contrast in the final image. As each of the objectives of a microscope is different, it means that the sub-stage condenser will need to be adjusted when you change magnification.
Within the sub-stage condenser there are two component parts which are adjusted in setting up correct Koehler illumination. These are the centering screws and the diaphragm. As well as the field diaphragm, the sub-stage condenser also has a light controlling diaphragm which is adjusted by either a slide control or a knurled ring around the condenser similar to the field diaphragm. This diaphragm controls the angle of the light cone which is illuminating the specimen. As I alluded to in my article on Numerical Aperture (NA), the NA of the whole microscope system is not only determined by the objectives. Indeed, when the sub-stage condenser diaphragm is opened, this produces an increase in the NA of the whole instrument resulting in greater resolution, contrast and optimal illumination. The centering screws ensure that the light used to illuminate your slide is directly in the centre of the field of view, thereby offering uniform intensity across the specimen.
Five steps for Koehler Illumination
Step 1: Check that the lenses, eyepieces, objectives are clean
Before you start your imaging session always check to see that the lenses, eyepieces, objectives are clean. The cleaning of microscope lenses is covered in another article, but you should always use lens cleaning tissue and a recommended solvent. Place the slide on the stage, and turn on the lamp to its lowest setting.
Step 2: Close the field diaphragm
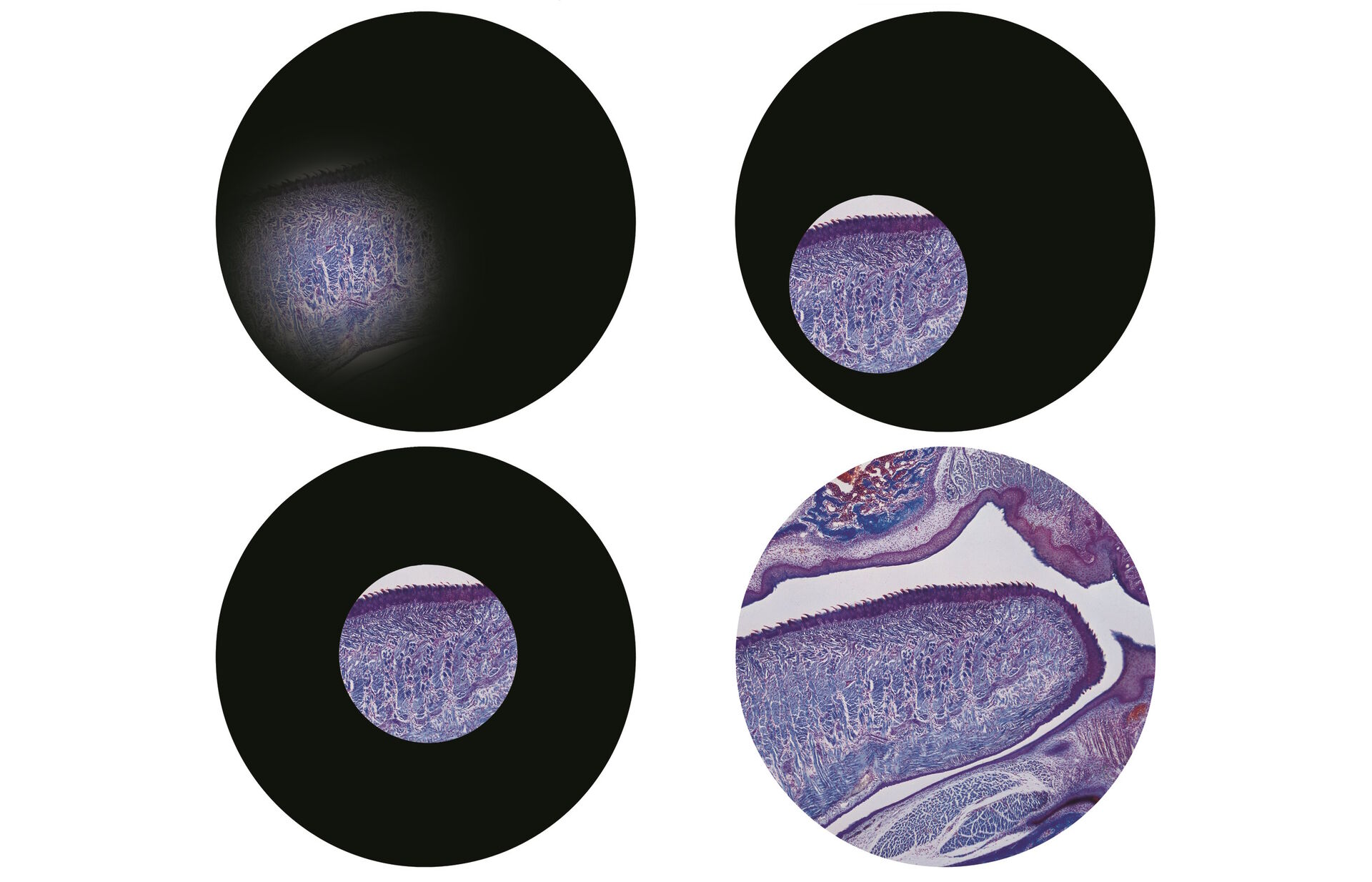
Focus your specimen, but remember if you are using an instrument in a shared facility, then the microscope settings may be completely misaligned.
Firstly, close down the field diaphragm which is either on the base of the instrument in an upright microscope, or above the condenser on an inverted microscope. When you look down the eyepieces, you’ll now just see a small circle of light on your slide (Figure 2).
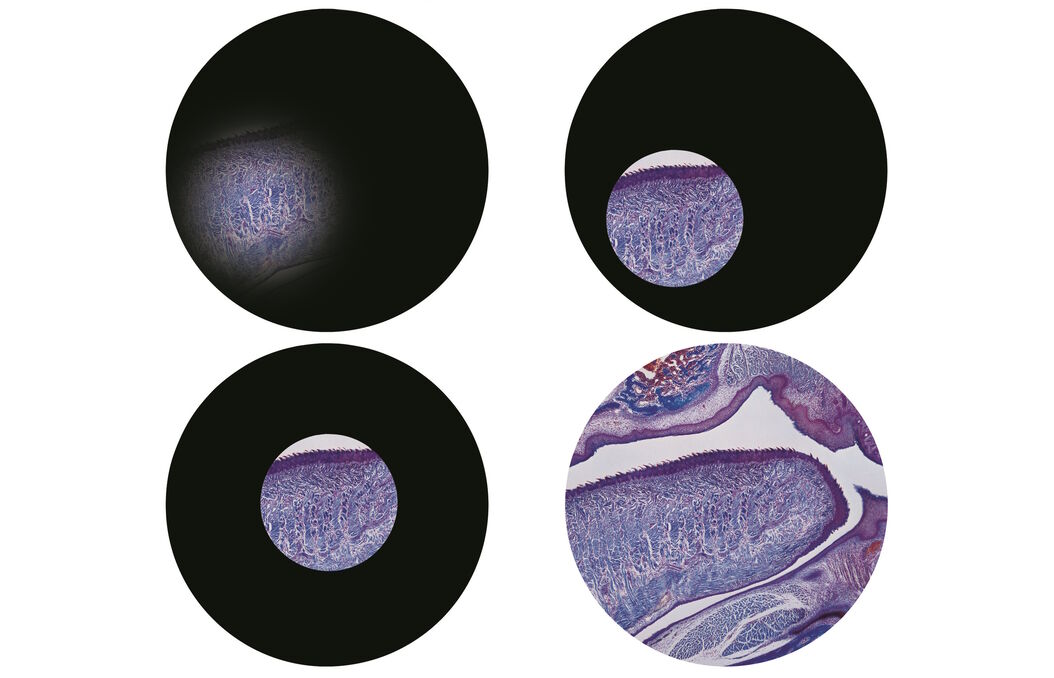
Step 3: Focus the condenser
Now focus the condenser. The adjustment wheels for the condenser are usually just in front of the focus wheels as you sit at the microscope. Move the condenser slowly up and down whilst looking down the eyepieces. You’ll see that the edge of the circle of light will go in and out of focus (and change from a red fringe to a blue fringe). Make the edges of the circle of light as sharp as you can (Figure 3).
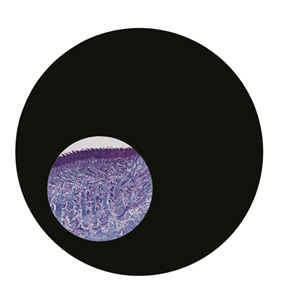
Step 4: Centre the condenser and open the field diaphragm
Once you have the sharp circle of light, look away from the eyepieces and have a look at the condenser component of the microscope. You’ll see the centering screws. You can either centre the condenser with the diaphragm closed down or almost fully open. I usually centre the condenser with the field diaphragm where I left it in step two above - almost fully closed down. Whilst holding the screws, look down the eyepieces once again. As you slowly turn the screws, you’ll see that the circle of light will move across the field of view. You should aim to have the circle in the middle of the view of your specimen (Figure 4).
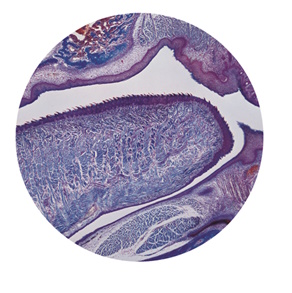
Once you have centred the condenser, open up the field diaphragm until the edges of the circle of light just disappear from the field of view (Figure 5).
Step 5: Final adjustments
The final adjustment is with the iris (or aperture) diaphragm. Just below the centering screws (or above on an inverted microscope) you’ll find another diaphragm similar to the main field diaphragm. Whilst looking down the eyepieces, open this fully, and then start closing it until the image ‘flare’ disappears. By closing the aperture diaphragm, you will increase the contrast of your specimen, but resolution will be decreased. You should find a balance depending on your eyes and the specimen you are viewing.
That’s it - five simple steps.
In summary - close down the diaphragm, focus the edges, centre the spot, open up the diaphragm and reduce flare.