What is fluorescence microscopy?
Fluorescence microscopy is done with an optical microscope where the illumination is provided by a laser, mercury-vapor or xenon arc lamp, or more recently LEDs (light emitting diodes). Additionally, there is an excitation and emission filter. The excitation filter transmits light which excites fluorescent dyes (fluorophores) or proteins in the specimen. The light emitted by fluorophores pass through the emission filter to reach the detector. A filter only allows through light with a distinct wavelength.
Stokes Shift
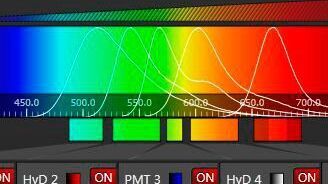
A trait of fluorescence is the Stokes shift: the energy difference between excitation and emission photons. Emitted photons have longer wavelengths than excitation photons as energy is released to the surroundings. The wavelength shift often depends on the fluorophore.
Related articles on the basic principles of fluorescence
An Introduction to Fluorescence
Epi-Illumination Fluorescence and Reflection-Contrast Microscopy
Basic Principles of Luminescence
Overview of Fluorescent Dyes in terms of Applications and Properties
Fluorescent Proteins - From the Beginnings to the Nobel Prize
Photoactivatable, Photoconvertible, and Photoswitchable Fluorescent Proteins
Basic operation of a fluorescence microscope
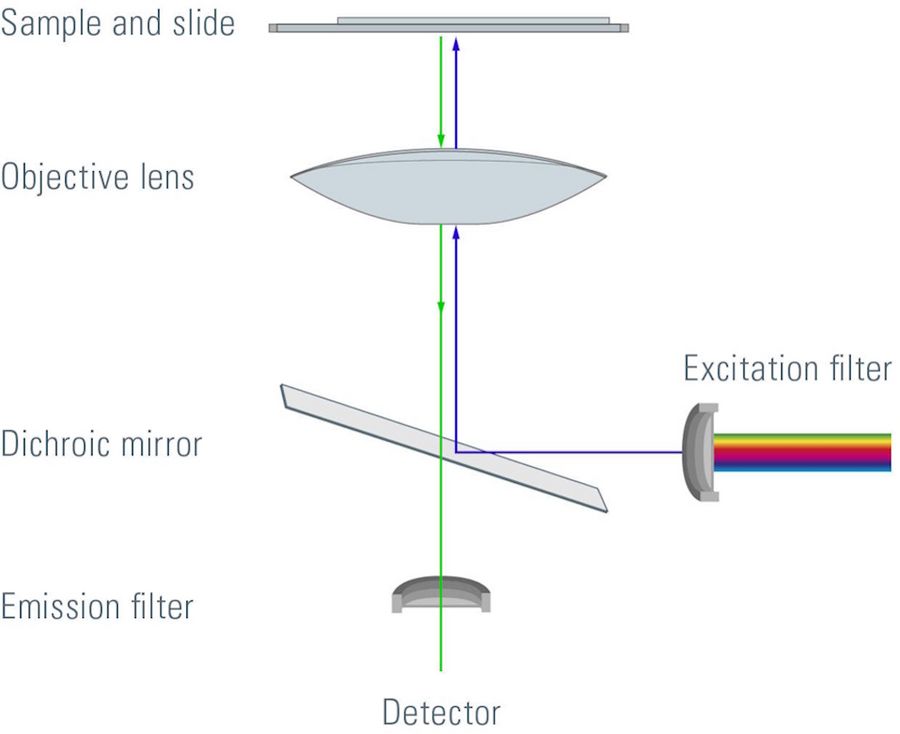
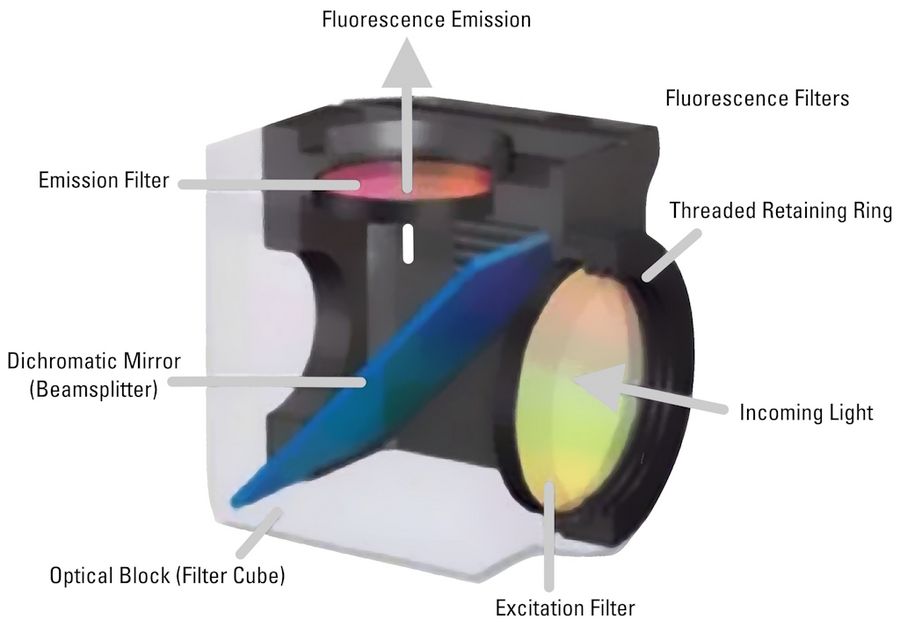
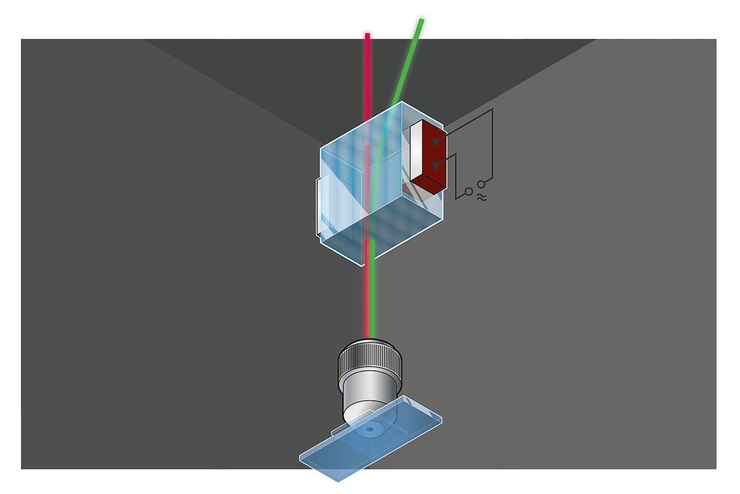
Most fluorescence microscopes use incident light fluorescence (also called epi-fluorescence) where the illumination and objective are on the same side of the specimen, so light is not passing through it. Besides the excitation and emission filter, there is also a dichroic mirror allowing only certain wavelengths to pass through. The filters and dichroic mirror are often together in a filter cube.
Light passes through the excitation filter, reflects from the dichroic mirror, and goes through the objective towards the specimen. Fluorophores in the specimen are excited and emit light going back through the objective. Only light with the appropriate wavelength pass the dichroic mirror and emission filter. Emitted photons are measured with a detector.
Filters for fluorescence
Band pass, long pass, and short pass filters are used for fluorescence microscopy. A band pass filter transmits only a specific range of wavelengths. A long pass filter transmits wavelengths above a certain value. A short pass filter transmits wavelengths below a certain value. Efficient sensing of emission light and full tunability is achieved with tunable spectral detection.
Related articles basic operation of a fluorescence microscope
Handbook of Optical Filters for Fluorescence Microscopy
Primary Beam Splitting Devices for Confocal Microscopes
Acousto Optics in True Confocal Spectral Microscope Systems
Confocal fluorescence microscopy
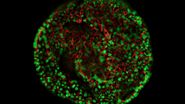
With confocal laser scanning microscopy (CLSM), limited specimen areas are observed with optical sectioning, as light signals from outside the focal plane are excluded. CLSM allows localization of biomolecules in cell organelles, cytoskeleton, or membranes.
Fluorescence lifetime imaging microscopy (FLIM) exploits how long a fluorophore remains in an excited state before relaxing and emitting a photon. Information about molecular spatial distributions and nanoscale environments can be obtained.
Special fluorescence techniques
- Total internal reflection (TIRF) microscopy enables fluorophores at a cell surface to be observed with evanescent light.
- With fluorescence recovery after photobleaching (FRAP), fluorophores in a small area are photobleached, then diffusion of unbleached molecules is observed.
- Fluorescence energy transfer (FRET) microscopy is useful for molecular interaction studies. A donor fluorophore can transfer energy to an acceptor if both are close enough together.
- Fluorescence in situ hybridization (FISH) uses fluorophores which bind to specific parts of a nucleic acid.
- Sub-resolution or super-resolution microscopy, circumventing the diffraction limit, is possible with stimulated emission depletion (STED), ground-state depletion (GSD), single molecule and ground state depletion followed by individual molecule return (GSDIM), photoactivated localization microscopy (PALM), and stochastic optical reconstruction microscopy (STORM).
Related articles on fluorescent techniques
A Guide to Fluorescence Lifetime Imaging Microscopy (FLIM)
Live-Cell Imaging Techniques
Total Internal Reflection Fluorescence (TIRF) Microscopy
Förster Resonance Energy Transfer (FRET)
A Guide to Super-Resolution
Fluorescence Correlation Spectroscopy (FCS)
How to Prepare your Specimen for Immunofluorescence Microscopy
FAQs Fluorescence Microscopy
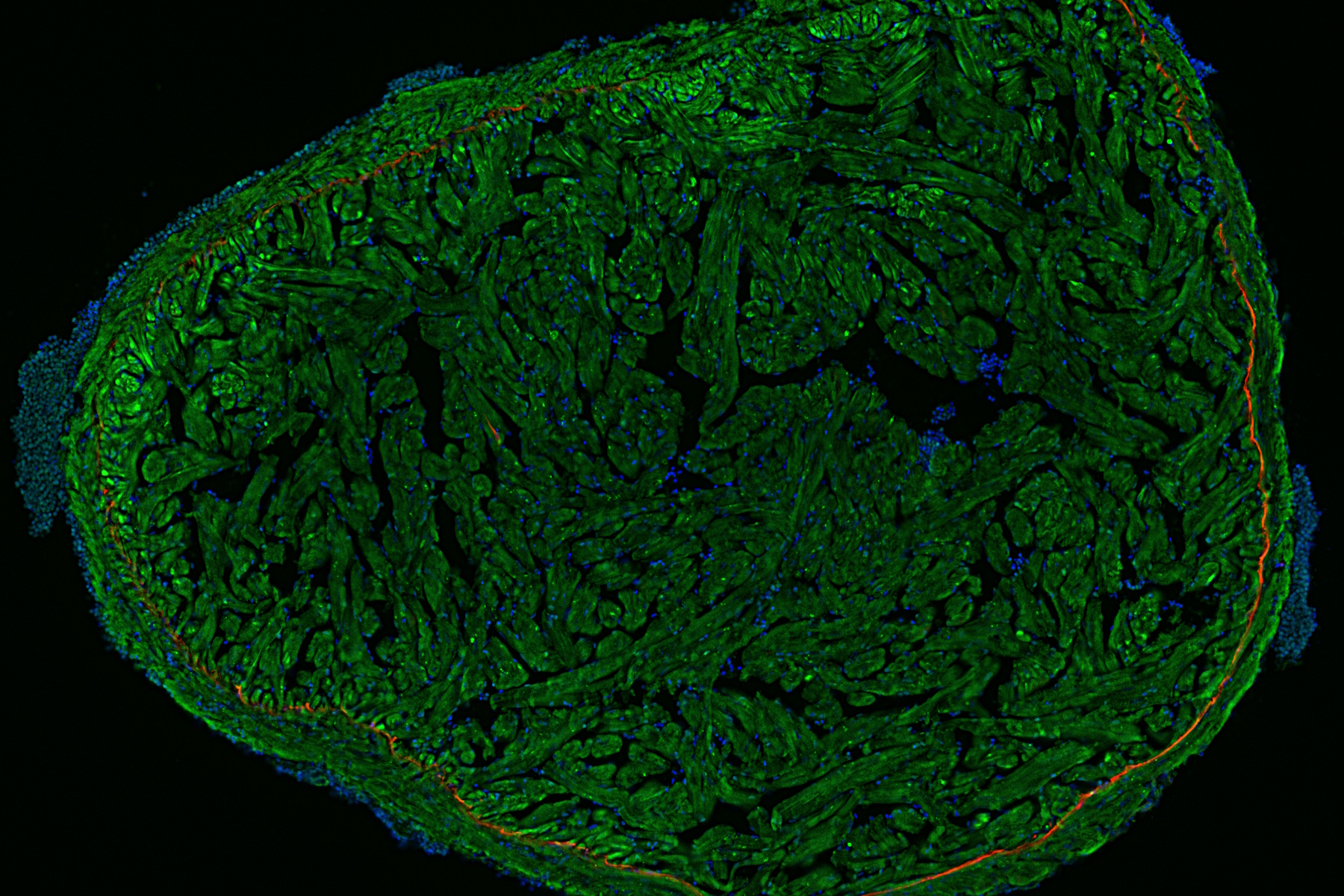
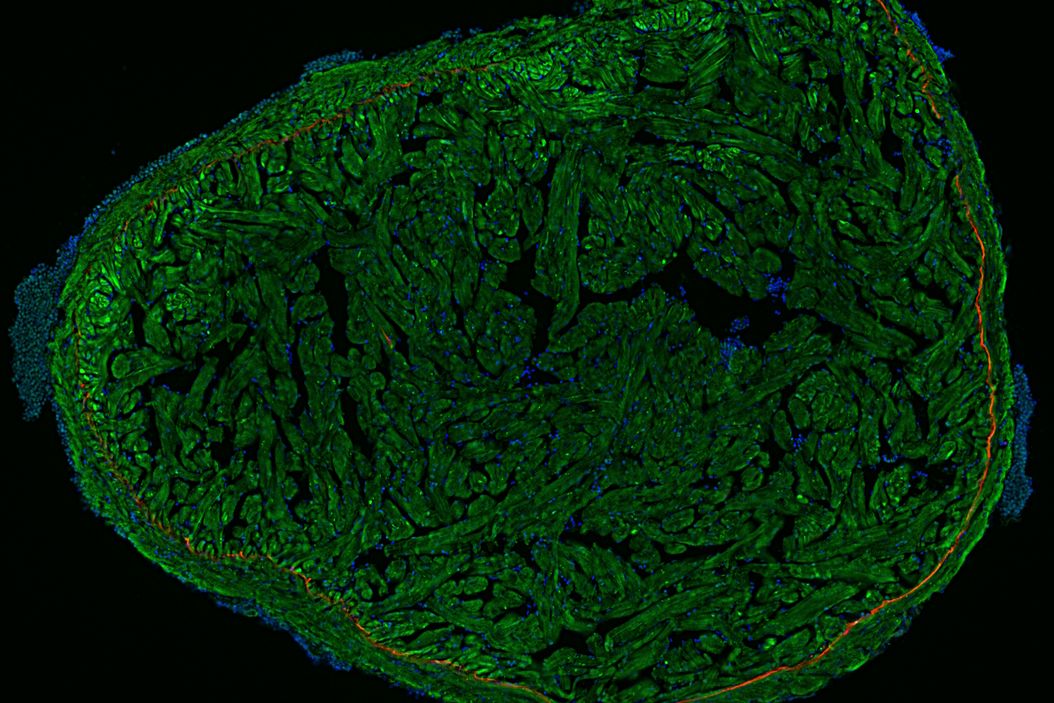
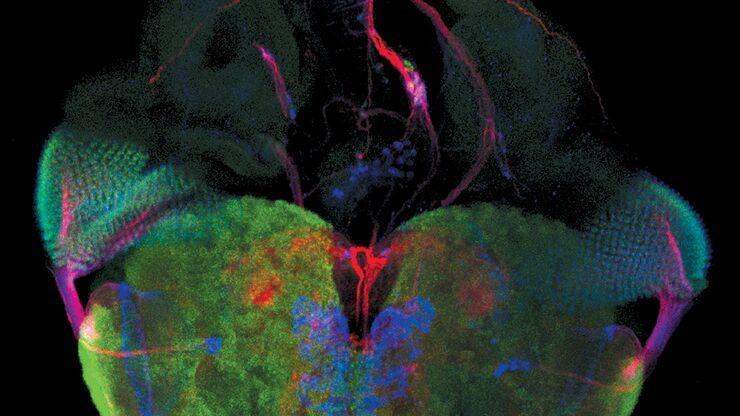
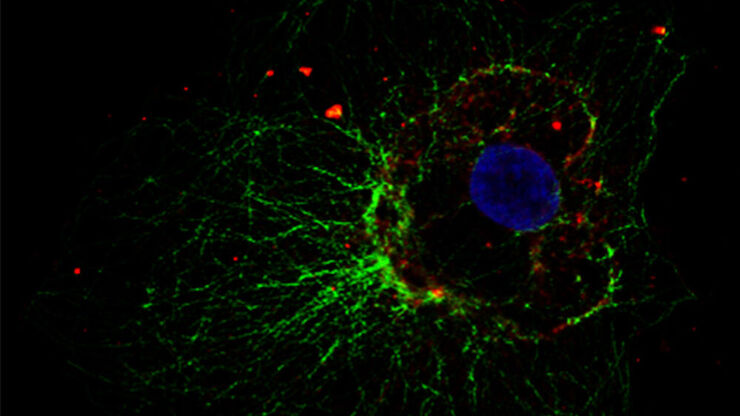
Fluorescence microscopy is often used to image specific structures of biological specimens, like cells and tissues. It requires fluorescent labels to be attached to targeted specimen structures, often with anti-bodies, or expression of fluorescent proteins in structures.
Fluorescence microscopy requires specimen structures to have fluorescent dyes, also called fluorophores or fluorochromes. These dye molecules become excited after absorbing light of a shorter wavelength and then emit light at a longer wavelength. The microscope needs a fluorescence light source as well as excitation and emission filters.
The setup and optics of brightfield and fluorescence microscopes are similar. However, in addition, fluorescence microscopes require a fluorescence light source and excitation and emission filters in order to see fluorescent dyes (fluorophores or fluorochromes) or proteins.
Common types of fluorescence microscopy are widefield fluorescence microscopy, confocal fluorescence microscopy, stereo fluorescence microscopy, total internal reflection (TIRF) microscopy, fluorescence recovery after photobleaching (FRAP) microscopy, fluorescence energy transfer (FRET) microscopy, fluorescence lifetime imaging microscopy (FLIM), among others.
The most commonly used fluorescence microscope is the widefield fluorescence compound microscope using incident light fluorescence illumination, also called epi-fluorescence illumination. To learn more about the development of this common fluorescence microscope, refer to this article on Learn & Share / Science Lab.